Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
EBV DNA load in the saliva of healthy people in south-eastern Poland
1
Aldent – Centre for Dentistry and Implantology, Lublin, Poland
2
Department of Virology with Viral Diagnostics Laboratory, Medical University, Lublin, Poland
3
Department of Medical Anthropology, Institute of Rural Health, Lublin, Poland
4
Faculty of Administration and Social Sciences, University of Economics and Innovation, Lublin, Poland
Corresponding author
Małgorzata Goździewska
Department of Medical Anthropology, Institute of Rural Health, Lublin, Jaczewskiego 2, 20-090, Lublin, Poland
Department of Medical Anthropology, Institute of Rural Health, Lublin, Jaczewskiego 2, 20-090, Lublin, Poland
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Epstein-Barr virus (EBV) is widespread in the world’s population. Transmitted mainly through saliva, the EBV virus infects the epithelial cells of the oropharynx, where it can replicate, establishing latency in B lymphocytes. Many authors analyse EBV viremia in saliva as a possible biomarker of various diseases. The aim of the study is to analyse the frequency and level of EBV viremia in the saliva of a healthy population of children and adults. The level of anti-EBV antibodies and a possible correlation between EBV viral load in saliva and antibody titres in serum were also examined.
Material and methods:
The study involved 66 healthy people, including 22 children aged 5–10 and 44 adults. EBV DNA was isolated from the saliva of all patients, and the presence and level of EBV DNA was detected using the qPCR method. Anti-EBV antibodies in serum were detected by ELISA.
Results:
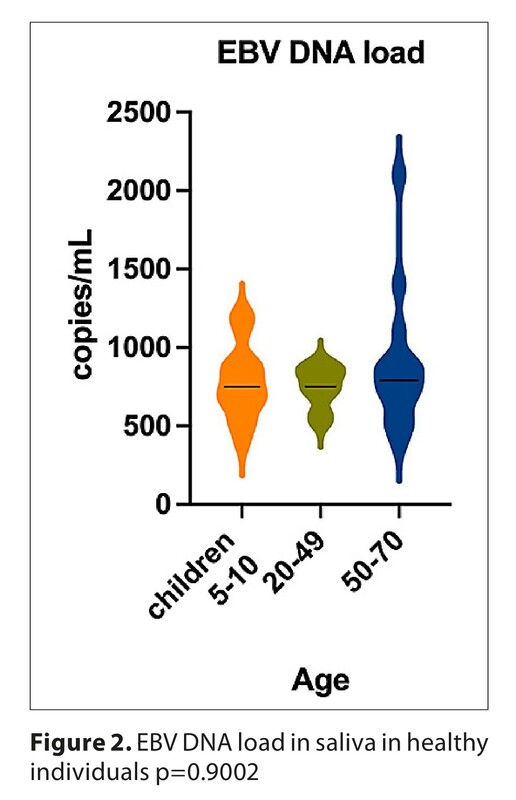
The higher percentage of positive results was detected in children aged 5–10 (81.8%) and a smaller percentage in adults aged 20–49 (75.0%), this difference was not statistically significant. EBV DNA was most often detected in adults aged 50–70 (85.7%). The mean level of EBVCA IgG antibodies was similar in both age groups. Similarly, no difference in EBNA-1 antibody titers was observed between younger and older groups of adult patients. In three healthy people, despite the presence of EBV DNA in saliva, no anti-EBVCA or EBNA-1 antibodies were detected.
Conclusions:
The prevalence of EBV DNA in the healthy population ranged from 75% – 85.7%. Additionally, no correlation was observed between the EBV load in saliva and the titer of tested antibodies in the serum of healthy people.
Epstein-Barr virus (EBV) is widespread in the world’s population. Transmitted mainly through saliva, the EBV virus infects the epithelial cells of the oropharynx, where it can replicate, establishing latency in B lymphocytes. Many authors analyse EBV viremia in saliva as a possible biomarker of various diseases. The aim of the study is to analyse the frequency and level of EBV viremia in the saliva of a healthy population of children and adults. The level of anti-EBV antibodies and a possible correlation between EBV viral load in saliva and antibody titres in serum were also examined.
Material and methods:
The study involved 66 healthy people, including 22 children aged 5–10 and 44 adults. EBV DNA was isolated from the saliva of all patients, and the presence and level of EBV DNA was detected using the qPCR method. Anti-EBV antibodies in serum were detected by ELISA.
Results:
The higher percentage of positive results was detected in children aged 5–10 (81.8%) and a smaller percentage in adults aged 20–49 (75.0%), this difference was not statistically significant. EBV DNA was most often detected in adults aged 50–70 (85.7%). The mean level of EBVCA IgG antibodies was similar in both age groups. Similarly, no difference in EBNA-1 antibody titers was observed between younger and older groups of adult patients. In three healthy people, despite the presence of EBV DNA in saliva, no anti-EBVCA or EBNA-1 antibodies were detected.
Conclusions:
The prevalence of EBV DNA in the healthy population ranged from 75% – 85.7%. Additionally, no correlation was observed between the EBV load in saliva and the titer of tested antibodies in the serum of healthy people.
REFERENCES (26)
1.
Current ICTV Taxonomy Release 2025 (MSL#41). Available online: https://ictv.global/taxonomy (accessed on 14 April 2026).
3.
Sharifipour S, Davoodi Rad K. Seroprevalence of Epstein-Barr virus among children and adults in Tehran, Iran. New Microbes New Infect. 2020;34:100641.
4.
Yu H, Robertson ES. Epstein–Barr Virus and Human Cancer: Epidemiol Pathogenesis Viruses. 2023;15(3):714.
5.
Huang W, Bai L, Tang H. Epstein-Barr virus infection: the micro and macro worlds. Virol J. 2023;20:220.
6.
Misra A, Speicher DJ, Luinstra K, et al. Self-collected oral flocked swabs to measure prevalence of Epstein–Barr Virus antibodies and DNA amongst university students. Diagn Microbiol Infect Dis. 2021;100(1):115295.
7.
Gieß RM, Pfuhl C, Behrens JR, et al. Epstein-Barr virus antibodies in serum and DNA load in saliva are not associated with radiological or clinical disease activity in patients with early multiple sclerosis. PLoS ONE 2017;12(4):e0175279.
8.
Portet Sulla VP, Kadi A, Mouna L, et al. Investigation of atypical serological profiles for Epstein–Barr virus (EBV). J Virol Methods. 2024;329:115002.
9.
Thorley-Lawson DA, Hawkins JB, Tracy SI, et al. The pathogenesis of Epstein-Barr virus persistent infection. Curr Opin Virol. 2013;3(3):227–32.
10.
Saito I, Nishimura S, Kudo I, et al. Detection of Epstein-Barr Virus and Human Herpes Virus Type 6 in Saliva from Patients with Lymphoproliferative Diseases by the Polymerase Chain Reaction. Arch Oral Biol. 1991;36(11):779–784.
11.
Dawson DR, Wang C, Danaher R, et al. Salivary Levels of Epstein-Barr Virus DNA Correlate with Subgingival Levels, Not Severity of Periodontitis. Oral Dis. 2009;15(8):554–559.
12.
Ling PD, Lednicky JA, Keitel WA, et al. The Dynamics of Herpesvirus and Polyomavirus Reactivation and Shedding in Healthy Adults: A 14-Month Longitudinal Study. J Infect Dis. 2003;187(10):1571–1580.
13.
Pow EHN, Law MYT, Tsang PCS, et al. Salivary Epstein-Barr Virus DNA Level in Patients with Nasopharyngeal Carcinoma Following Radiotherapy. Oral Oncol. 2011;47(9):879–882.
14.
He Y-Q, Zhou T, Yang D-W, et al. Prognostic Value of Oral Epstein–Barr Virus DNA Load in Locoregionally Advanced Nasopharyngeal Carcinoma. Front Mol Biosci. 2022;8:757644.
15.
Ikuta K, Satoh Y, Hoshikawa Y, et al. Detection of Epstein-Barr virus in salivas and throat washings in healthy adults and children. Microb Infect. 2000;2(2):115–120.
16.
Oral A, Onel M, Demirci M, et al. Investigation of the presence of Epstein-Barr virus in patients who had Oral Lichen Planus and Oral Lichenoid Contact Lesions with Real-time PCR method in serum, tissue and saliva samples. North Clin Istanb. 2024;11(6):569–574.
17.
Zheng XH, Zhou T, Li XZ, et al. Evaluation of Epstein–Barr virus DNA methylation and load in saliva in the management of individuals at high risk of nasopharyngeal carcinoma. Biomark Res. 2026;14:17.
18.
Grimm-Geris J, Dunmire S, Duval L, et al. Screening for Epstein–Barr virus (EBV) infection status in university freshmen: acceptability of a gingival swab method. Epidemiol Infect. 2019;147.
19.
Hjalgrim HFJ, Melbye M. The epidemiology of EBV and its association with malignant disease. In: Arvin A, Campadelli-Fiume G, Mocarski E, ed. Human herpesviruses: biology, therapy, and immunoprophylaxis. Cambridge: Cambridge University Press; 2007. Chapter 53.
20.
Balfour HHJr, Sifakis F, Sliman JA, et al. Age-specific prevalence of Epstein–Barr virus infection among individuals aged 6–19 years in the United States and factors affecting its acquisition. J Infect Dis. 2013;208:1286–1293.
21.
Dowd JB, Palermo T, Brite J, et al. Seroprevalence of Epstein–Barr virus infection in US children ages 6–19, 2003–2010. PloS One 2013;8(5):e64921.
22.
Levine H, Balicer RD, Rozhavski V, et al. Seroepidemiology of Epstein−Barr virus and cytomegalovirus among Israeli male young adults. Ann Epidemiol. 2012;22:783–788.
23.
Condon LM, Cederberg LE, Rabinovitch MD, et al. Age-specific prevalence of Epstein–Barr virus infection among Minnesota children: effects of race/ethnicity and family environment. Clin Infect Dis. 2014;59:501–508.
24.
Mahler JV, Bilodeau PA, Anderson M, et al. A threshold in anti-EBNA-1 antibody titers distinguishes salivary EBV shedders from non-shedders. J Infect Dis. 2026, jiag010.
25.
Paradowski K, Góralczyk M, Drop B, Jarosz M, et al. Epstein–Barr Virus Load in the Saliva of Patients with Oropharyngeal Cancer—Could It Have Prognostic Significance? Viruses 2025;17:1523.
26.
Nyeo SS, Cumming EM, Burren OS, et al. Population-scale sequencing resolves determinants of persistent EBV DNA. Nature 2026;650:664–672.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.