Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Analysis of the concentration of selected serological parameters in patients undergoing
antibiotic treatment of Lyme disease and assessment of their potential application in the
control of the therapy effectiveness – pilot study
1
Faculty of Health Sciences, Pope John Paul II State School of Higher Education in Biała Podlaska, Poland
2
Department of Infectious Diseases, Medical University, Lublin, Poland
3
Witold Chodźko Institute of Rural Heath, Lublin, Poland
4
Department of Microbiological Diagnostics, Clinical Hospital No. 1, Lublin, Poland
5
Innovation Research Centre, Pope John Paul II State School of Higher Education in Biała Podlaska, Poland
Corresponding author
Małgorzata Tokarska-Rodak
Faculty of Health Sciences, Pope John Paul II State School of Higher Education in Biala Podlaska, Sidorska 95-97, 21-500, Biała Podlaska, Poland
Faculty of Health Sciences, Pope John Paul II State School of Higher Education in Biala Podlaska, Sidorska 95-97, 21-500, Biała Podlaska, Poland
Ann Agric Environ Med. 2021;28(4):605-611
KEYWORDS
TOPICS
ABSTRACT
Objective:
The aim of the study was to analyze the concentrations of some serological parameters in patients undergoing antibiotic therapy for LD, and to assess the potential use of IgG anti-C6VlsE in the assessment of treatment effectiveness.
Material and methods:
The study involved 21 patients diagnosed with LD. To determine the levels of anti-C6VlsE IgG, metalloproteinases and cytokines, blood was collected three times: before treatment, immediately after treatment, and 3–4 months after treatment.
Results:
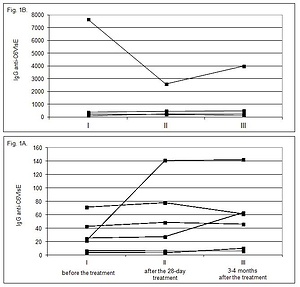
Immediately after the antibiotic therapy, all the patients in the study group declared improvement in their health. At 3–4 months post-treatment, 50% of patients reported recurrence of the disease symptoms. The anti-C6VlsE IgG concentrations at the analyzed time points exhibited varied dynamics: in 15% of patients, a decrease in the concentration of anti-C6VlsE was observed in the three consecutive serum samples; an increase of anti-C6VlsE IgG was detected in 35% of cases; in 15% of the subjects, the concentration of anti-C6VlsE IgG increased immediately after the antibiotic therapy and declined after 3–4 months, whereas a reverse situation was noted in 10% of the patients.
Conclusions:
The presence of high concentrations of anti-C6VlsE IgG is associated with appearance of response to Borreliella spp. infection, which suggests that the parameter can be used in the diagnosis of the active form of borreliosis. However, from a practical point of view, a marker of successful elimination of infection should indicate the effectiveness or failure of antibiotic therapy within a relatively short post-treatment period. The anti-C6VlsE IgG antibodies do not seem to satisfy this provision.
The aim of the study was to analyze the concentrations of some serological parameters in patients undergoing antibiotic therapy for LD, and to assess the potential use of IgG anti-C6VlsE in the assessment of treatment effectiveness.
Material and methods:
The study involved 21 patients diagnosed with LD. To determine the levels of anti-C6VlsE IgG, metalloproteinases and cytokines, blood was collected three times: before treatment, immediately after treatment, and 3–4 months after treatment.
Results:
Immediately after the antibiotic therapy, all the patients in the study group declared improvement in their health. At 3–4 months post-treatment, 50% of patients reported recurrence of the disease symptoms. The anti-C6VlsE IgG concentrations at the analyzed time points exhibited varied dynamics: in 15% of patients, a decrease in the concentration of anti-C6VlsE was observed in the three consecutive serum samples; an increase of anti-C6VlsE IgG was detected in 35% of cases; in 15% of the subjects, the concentration of anti-C6VlsE IgG increased immediately after the antibiotic therapy and declined after 3–4 months, whereas a reverse situation was noted in 10% of the patients.
Conclusions:
The presence of high concentrations of anti-C6VlsE IgG is associated with appearance of response to Borreliella spp. infection, which suggests that the parameter can be used in the diagnosis of the active form of borreliosis. However, from a practical point of view, a marker of successful elimination of infection should indicate the effectiveness or failure of antibiotic therapy within a relatively short post-treatment period. The anti-C6VlsE IgG antibodies do not seem to satisfy this provision.
REFERENCES (27)
1.
Bamm VV, Ko JT, Mainprize IL, et al. Lyme Disease Frontiers: Reconciling Borrelia Biology and Clinical Conundrums. Pathogens. 2019; 8(4): 299. https://doi.org/10.3390/pathog....
2.
National Institute for Health and Care Excellence. Lyme disease. NICE guideline (NG95). Published date: 11 April 2018, https://www.nice.org.uk/guidan... (access: 2020.10.01).
3.
Pancewicz SA, Garlicki AM, Moniuszko-Malinowska A, et al. Diagnosis and treatment of tick-borne diseases recommendations of the Polish Society of Epidemiology and Infectious Diseases. Przegl Epidemiol. 2015; 69(2): 309–316.
4.
Chmielewski T, Dunaj J, Gołąb E, et al. Diagnostyka laboratoryjna chorób odkleszczowych. Krajowa Izba Diagnostów Laboratoryjnych, Warszawa: Rekomendacje Grupy Roboczej 2014, 1–50. https://kidl.org.pl/get-file/1... (access: 2020.09.16).
5.
Krzemień PJ. Role of VIsE/C6 antigen as a marker for early Lyme borreliosis diagnosis and monitoring the effectiveness of its treatment. Health Probl Civiliz. 2017; 11(2): 87–92. https://doi.org/10.5114/hpc.20....
6.
Zajkowska JM, Kondrusik M, Pancewicz SA, et al. Comparison of test with antigen VlsE (C6) with tests with recombinant antigens in patients with Lyme borreliosis. Pol Merkur Lek. 2007; 23(134): 95–99.
7.
Cinco M, Murgia R. Evaluation of the C6 enzyme-linked immuno-adsorbent assay for the serodiagnosis of Lyme borreliosis in north-eastern Italy. New Microbiol. 2006; 29(2): 139–141.
8.
Marangoni A, Sambri V, Accardo S, et al. A Decrease in the Immuno-globulin G Antibody Response against the VlsE Protein of Borrelia burgdorferi Sensu Lato Correlates with the Resolution of Clinical Signs in Antibiotic-Treated Patients with Early Lyme Disease. Clin Vaccine Immunol. 2006; 13(4): 525–529. https://doi.org/10.1128/CVI.13....
9.
Fleming RV, Marques AR, Klempner MS, et al. Pre-treatment and post-treatment assessment of the C(6) test in patients with persistent symptoms and a history of Lyme borreliosis. Eur J Clin Microbiol Infect Dis. 2004; 23(8): 615–618. https://doi.org/10.1007/s10096....
10.
Petnicki-Ocwieja T, Kern A. Mechanisms of Borrelia burgdorferi internalization and intracellular innate immune signaling. Frontiers in Cellular and Infection Microbiology 2014; 4(175): 1–7. https://doi.org/10.3389/fcimb.....
11.
Petnicki-Ocwieja T, Chung E, Acosta DI, et al. TRIF mediates Toll-like receptor 2-dependent inflammatory responses to Borrelia burgdorferi. Infect Immun. 2013; 81: 402–410. https://doi.org/10.1128/IAI.00....
12.
Baszczuk A, Kopczyński Z, Thielemann A, et al. Evaluation of concentrations of metalloproteinase 2 (MMP-2) and metalloproteinase 9 (MMP-9) in serum of patients with primary hypertension. Forum Zaburzeń Metabolicznych 2015; 6(2): 74–84.
13.
Sulka KB, Strle K, Crowley JT, et al. Lyme Disease-Associated IgG4 autoantibodies correlate with synovial pathology in antibiotic-refractory Lyme arthritis. Arthritis Rheumatol. 2018; 70(11): 1835–1846. https://doi.org/10.1002/art.40....
14.
Strle K, Sulka KB, Pianta A, et al. T-Helper 17 cell cytokine responses in Lyme Disease correlate with Borrelia burgdorferi antibodies during early infection and with autoantibodies late in the illness in patients with antibiotic-refractory Lyme arthritis. CID. 2017; 64(7): 930–8 https://doi.org/10.1093/cid/ci....
15.
Crowley JT, Strle K, Drobin EE, et al. Matrix metalloproteinase-10 is a target of T and B cell responses that correlate with synovial pathology in patients with antibiotic-refractory Lyme arthritis. J Autoimmun. 2016; 69: 24–37. https://doi.org/10.1016/j.jaut....
16.
Philipp MT, Wormser GP, Marques AR, et al. A Decline in C6 antibody titer occurs in successfully treated patients with culture-confirmed early localized or early disseminated Lyme borreliosis. Clin Diagn Lab Immunol. 2005; 12(9): 1069–1074. https://doi.org/10.1128/CDLI.1....
17.
Pegalajar-Jurado A, Schriefer ME, Welch RJ, et al. Evaluation of Modified Two-Tiered Testing Algorithms for Lyme disease laboratory diagnosis using Well-Characterized Serum Samples. J Clin Microbiol. 2018; 56: e01943–17. https://doi.org/10.1128/JCM.01....
18.
Zhao H, Dai X, Han X, et al. Borrelia burgdorferi basic membrane protein a initiates proinflammatory chemokine storm in THP 1-derived macrophages via the receptors TLR1 and TLR2. Biomedicine & Pharmacotherapy 2019; 115: 108874. https://doi.org/10.1016/j.biop....
19.
Aucott JN, Soloski MJ, Rebman AW, et al. CCL19 as a chemokine risk factor for Posttreatment Lyme Disease Syndrome: a prospective clinical cohort study. Clin Vaccine Immunol. 2016; 23(9): 757–766. https://doi.org/10.1128/CVI.00....
20.
Steere AC, Strle F, Wormser GP. Lyme borreliosis. Nat Rev Dis Primers. 2016; 2: 16090. https://doi.org/10.1038/nrdp.2....
21.
Liu M, Guo S, Hibbert JM, et al. CXCL10/IP-10 in infectious diseases pathogenesis and potential therapeutic implications. Cytokine Growth Factor Rev. 2011; 22(3): 121–130. https://doi.org/10.1016/j.cyto....
22.
Fallahi P, Elia G, Bonatti A. Interferon-?-induced protein 10 in Lyme disease. Clin Ter. 2017; 168(2): e146–150. https://doi.org/10.7417/CT.201....
23.
Codolo G, Bossi F, Durigutto P, et al. Orchestration of inflammation and adaptive immunity in Borrelia burgdorferi induced arthritis by neutrophil-activating protein A. Arthritis Rheum. 2013; 65: 1232–42. https://doi.org/10.1002/art.37....
24.
Hoversten K, Bartlett MA. Diagnosis of a tick-borne coinfection in a patient with persistent symptoms following treatment for Lyme disease. BMJ Case Rep. 2018; 2018: bcr2018225342. https://doi.org/10.1136/bcr-20....
25.
Velušček M, Blagus R, Cerar Kišek T. Antibiotic Use and Long-Term Outcome in Patients with Tick-Borne Encephalitis and Co-Infection with Borrelia Burgdorferi Sensu Lato in Central Europe. A Retrospective Cohort Study. J Clin Med. 2019; 8(10): 1740. https://doi.org/10.3390/jcm810....
26.
Dunaj J, Moniuszko-Malinowska A, Swiecicka I, et al. Tick-borne infections and co-infections in patients with non-specific symptoms in Poland. Adv Med Sci. 2018; 63(1): 167–172. https://doi.org/10.1016/j.advm....
27.
Moniuszko A, Dunaj J, Święcicka I, et al. Co-infections with Borrelia species, Anaplasma phagocytophilum and Babesia spp. in patients with tick-borne encephalitis. Eur J Clin Microbiol Infect Dis. 2014; 33(10): 1835–1841. https://doi.org/10.1007/s10096....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.