Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Analysis of selected serological parameters in patients with diagnosed Lyme borreliosis and in
seropositive patients with no clinical symptoms
1
Faculty of Health Sciences, Pope John Paul II State School of Higher Education, Biala Podlaska, Poland
2
Institute of Rural Health, Lublin, Poland
3
Department of Infectious Diseases, Skubiszewski Medical University, Lublin, Poland
4
Department of Microbiological Diagnostics Clinical Hospital No. 1, Lublin, Poland
Corresponding author
Anna Pańczuk
Faculty of Health Sciences, Pope John Paul II State School of Higher Education in Biala Podlaska, Sidorska 95/97, 21-500, Biała Podlaska, Poland
Faculty of Health Sciences, Pope John Paul II State School of Higher Education in Biala Podlaska, Sidorska 95/97, 21-500, Biała Podlaska, Poland
Ann Agric Environ Med. 2021;28(3):397-403
KEYWORDS
TOPICS
ABSTRACT
Objectives:
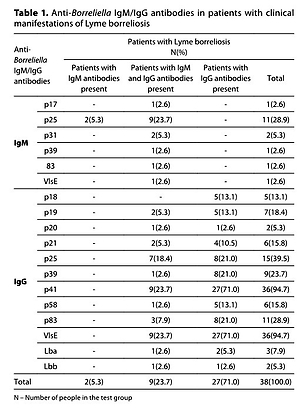
The aim of the study was to analyze some metalloproteinases, cytokines, and chemokines in LB patients and healthy seropositive subjects. The presence of IgM/IgG antibodies against specific Borreliella antigens was analyzed in the presence or absence of clinical manifestations of LB.
Material and methods:
The study involved 38 patients diagnosed with LB and arthralgia and/or arthritis symptoms, and 57 foresters presenting no clinical symptoms of LB. The ELISA test was applied for general screening of anti-Borreliella IgM/IgG. Western blot was used for confirmatory diagnosis of LB for the positive and borderline results. Serum IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF, IL-8, CCL5, CXCL9/MIG, CCL2/MCP-1, CXCL10/IP-10 concentrations were measured with the use of the Human Cytometric CBA test. The concentration of MMP-2 and MMP-9 in the serum was determined with the use of ELISA tests.
Results:
Analysis of the cytokines and chemokines revealed that only the concentration of IL-2 was significantly higher (2.4 pg/m; p=0.00641) in patients with LB symptoms than in the seropositive individuals (0.4 pg/ml). The MMP2 concentration was significantly higher (233.3 ng/ml; p=0.00294) in patients with clinical manifestations of LB than in those occupationally exposed to tick bites, but did not have anti-Borreliella antibodies (192.0 ng/ml).
Conclusions:
The presence of IgG antibodies against a number of Borreliella antigens and the differences in the IL-2 and MMP2 levels in seropositive or seronegative individuals and symptomatic LB patients, may indicate differences in the intensity of the immune response to the infection and, consequently, may induce development of clinical manifestations of the disease in seropositive and seronegative individuals.
The aim of the study was to analyze some metalloproteinases, cytokines, and chemokines in LB patients and healthy seropositive subjects. The presence of IgM/IgG antibodies against specific Borreliella antigens was analyzed in the presence or absence of clinical manifestations of LB.
Material and methods:
The study involved 38 patients diagnosed with LB and arthralgia and/or arthritis symptoms, and 57 foresters presenting no clinical symptoms of LB. The ELISA test was applied for general screening of anti-Borreliella IgM/IgG. Western blot was used for confirmatory diagnosis of LB for the positive and borderline results. Serum IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF, IL-8, CCL5, CXCL9/MIG, CCL2/MCP-1, CXCL10/IP-10 concentrations were measured with the use of the Human Cytometric CBA test. The concentration of MMP-2 and MMP-9 in the serum was determined with the use of ELISA tests.
Results:
Analysis of the cytokines and chemokines revealed that only the concentration of IL-2 was significantly higher (2.4 pg/m; p=0.00641) in patients with LB symptoms than in the seropositive individuals (0.4 pg/ml). The MMP2 concentration was significantly higher (233.3 ng/ml; p=0.00294) in patients with clinical manifestations of LB than in those occupationally exposed to tick bites, but did not have anti-Borreliella antibodies (192.0 ng/ml).
Conclusions:
The presence of IgG antibodies against a number of Borreliella antigens and the differences in the IL-2 and MMP2 levels in seropositive or seronegative individuals and symptomatic LB patients, may indicate differences in the intensity of the immune response to the infection and, consequently, may induce development of clinical manifestations of the disease in seropositive and seronegative individuals.
ACKNOWLEDGEMENTS
We are grateful to Adam Szepeluk for technical assistance
in statistical analysis.
FUNDING
This work was funded by the Pope John Paul II State School of
Higher Education in Biala Podlaska; “Analysis of the immune
response to Borrelia burgdorferi infection in patients treated
for Lyme disease” [grant number IV,2016]; “Analysis of the
secretion of selected cytokines and chemokines in patients
with Lyme borreliosis” [grant number S_11_17, 20017].
REFERENCES (41)
1.
Barbour AG, Adeolu M, Gupta RS. Division of the genus Borrelia into two genera (corresponding to Lyme disease and relapsing fever groups) reflects their genetic and phenotypic distinctiveness and will lead to a better understanding of these two groups of microbes (Margos et al. (2016) There is inadequate evidence to support the division of the genus Borrelia. Int J Syst Evol Microbiol. doi: 10.1099/ijsem.0.001717). Int J Syst Evol Microbiol. 2017; 67(6): 2058–2067. https://doi.org/10.1099/ijsem.....
2.
Stanek G, Strle F. Lyme borreliosis – from tick bite to diagnosis and treatment. FEMS Microbiol Rev. 2018; 42(3): 233–258. https://doi.org/10.1093/femsre....
3.
Stanek G, Wormser GP, Gray J, Strle F. Lyme borreliosis. Lancet. 2012; 379: 461–473. https://doi.org/10.1016/S0140-....
4.
Steere AC, Strle F, Wormser GP, Hu LT, Branda JA, Havius JW, et al. Lyme borreliosis. Nat Rev Dis Primers. 2016; 2: 16090. https://doi.org/10.1038/nrdp.2....
5.
Stanek G. Lyme borreliosis, ticks and Borrelia species. Wien Klin Wochenschr. 2018; 130: 459–462. https://doi.org/10.1007/s00508....
6.
Eldin C, Raffetin A, Bouiller K, Hansmann Y, Roblot F, Raoult D, et al. Review of European and American guidelines for the diagnosis of Lyme borreliosis. Med Mal Infect. 2019; 49(2): 121–132. https://doi.org/10.1016/j.medm....
7.
Pancewicz SA, Garlicki AM, Moniuszko-Malinowska A, Zajkowska J, Kondrusik M, Grygorczuk S, et al. Polish Society of Epidemiology and Infectious Diseases. Diagnosis and treatment of tick-borne diseases recommendations of the Polish Society of Epidemiology and Infectious Diseases. Przegl Epidemiol. 2015; 69(2): 309–316.
8.
Stanek G, Fingerle V, Hunfeld KP, Jaulhac B, Kaiser R, Krause A, et al. Lyme borreliosis: clinical case definitions for diagnosis and management in Europe. Clin Microbiol Infect. 2011; 17: 69–79. https://doi.org/10.1111/j.1469....
9.
Dehnert M, Fingerle V, Klier C, Talaska T, Schlaud M, Krause G, et al. Seropositivity of Lyme Borreliosis and Associated Risk Factors: A Population-Based Study in Children and Adolescents in Germany (KiGGS). PLoS ONE 2012; 7(8): e41321. https://doi.org/10.1371/journa....
10.
Ekerfelt C, Forsberg P, Svenvik M, Roberg M, Bergstrom S, Ernerudh J. Asymptomatic Borrelia-seropositive individuals display the same incidence of Borrelia-specific interferon-gamma (IFN-g)-secreting cells in blood as patients with clinical Borrelia infection. Clin Exp Immunol. 1999; 115: 498–502. https://doi.org/10.1046/j.1365....
11.
Hjetland R, Nilsen RM, Grude N, Ulvestad E. Seroprevalence of antibodies to Borrelia burgdorferi sensu lato in healthy adults from western Norway: risk factors and methodological aspects. APMIS. 2014; 122(11): 1114–1124. https://doi.org/10.1111/apm.12....
12.
Hristea A, Hristescu S, Ciufecu C, Vasile A. Seroprevalence of Borrelia burgdorferi in Romania. Eur J Epidemiol. 2001; 17: 891–896. https://doi.org/10.1023/a:1015....
13.
Pańczuk A, Kozioł-Montewka M, Tokarska-Rodak M. Exposure to ticks and seroprevalence of Borrelia burgdorferi among a healthy young population living in the area of southern Podlasie, Poland. Ann Agric Environ Med. 2014; 21(3): 512–517. https://doi.org/10.5604/123219....
14.
Skogman BH, Hellberg S, Ekerfelt C, Jenmalm MC, Forsberg P, Ludvigsson J, et al. Adaptive and innate immune responsiveness to Borrelia burgdorferi sensu lato in exposed asymptomatic children and children with previous clinical Lyme borreliosis. Clin Dev Immunol. 2012; 2012: 294587. https://doi.org/10.1155/2012/2....
15.
Steere AC, Sikand VK, Schoen RT, Nowakowski J. Asymptomatic infection with Borrelia burgdorferi. Clin Infect Dis. 2003; 37: 528–532. https://doi.org/10.1086/376914.
16.
Tomao P, Ciceroni L, D’Ovidio MC, De Rosa M, Vonesch N, Iavicoli S, et al. Prevalence and incidence of antibodies to Borrelia burgdorferi and to tick-borne encephalitis virus in agricultural and forestry workers from Tuscany, Italy. Eur J Clin Microbiol Infect Dis. 2005; 24: 457–463. https://doi.org/10.1007/s10096....
17.
Wilhelmsson P, Fryland L, Lindblom P, Sjöwall J, Ahlm C, Berglund J, et al. A prospective study on the incidence of Borrelia burgdorferi sensu lato infection after a tick bite in Sweden and on the Aland Islands, Finland (2008–2009). Ticks Tick Borne Dis. 2016; 7: 71–79. https://doi.org/10.1016/j.ttbd....
18.
Wilking H, Fingerle V, Klier C, Thamm M, Stark K. Antibodies against Borrelia burgdorferi sensu lato among Adults, Germany, 2008–2011. Emerg Infect Dis. 2015; 21(1): 107–110. https://doi.org/10.3201/eid210....
19.
Zákutná Ľ, Dorko E, Rimárová K, Kizeková M. Pilot Cross-Sectional Study of Three Zoonoses (Lyme Disease, Tularaemia, Leptospirosis) among Healthy Blood Donors in Eastern Slovakia. Cent Eur J Public Health. 2015; 23(2): 100–106. https://doi.org/10.21101/cejph....
20.
Pancewicz S, Rutkowski R, Rutkowski K, Zajkowska JM, Kondrusik M. Immunopathology of Lyme arthritis. Pol Merkur Lek. 2007; 23(134): 141–144.
21.
Zhao Z, Fleming R, McCloud B, Klempner MS. CD14 Mediates Cross Talk between Mononuclear Cells and Fibroblasts for Upregulation of Matrix Metalloproteinase 9 by Borrelia burgdorferi. Infect Immun. 2007; 75(6): 3062–3069. https://doi.org/10.1128/IAI.00....
22.
Zajkowska JM, Pancewicz SA, Grygorczuk S, Kondrusik M, Moniuszko A, Lakwa K. Neuroborreliosis – some aspects of pathogenesis, diagnosis and treatment. Pol Merkur Lek. 2008; 24(143): 453–457.
23.
Baszczuk A, Kopczyński Z, Thielemann A, Musialik K, Kopczyński J, Bielawska L, et al. Evaluation of concentrations of metalloproteinase 2 (MMP-2) and metalloproteinase 9 (MMP-9) in serum of patients with primary hypertension. Forum Zab Metabol. 2015; 6(2): 74–84.
24.
Lipka D, Boratyński J. Metalloproteinases. Structure and function. Postepy Hig Med Dosw. (online) 2008; 62: 328–336. http://www.phmd.pl/api/files/v... (accessed 15 March 2018).
25.
Chmielewski T, Dunaj J, Gołąb E, Gut W, Horban A, Pancewicz S, et al. Diagnostyka laboratoryjna chorób odkleszczowych. Krajowa Izba Diagnostów Laboratoryjnych, Warszawa, 2014. https://kidl.org.pl/file/file/... (access: 2018.03.02).
26.
Shkilna M, Tokarska-Rodak M, Pańczuk A, Smiyan S, Andreychyn M, Klishch I, et al. The use of anti-C6VlsE IgG in the assessment of the effectivness of Lyme disease treatment – a preliminary report. Health Probl Civiliz. 2019; 13(1): 83–91. https://doi.org/10.5114/hpc.20....
27.
Dankiewicz K, Tokarska-Rodak M, Weiner M. Prophylaxis of Lyme borreliosis and rural residents’ awareness. Health Probl Civiliz. 2018; 12(1): 1–6. https://doi.org/10.5114/hpc.20....
28.
Krzemień PJ. Role of VlsE/C6 antigen as a marker for early Lyme borreliosis diagnosis and monitoring the effectiveness of its treatment. Health Probl Civiliz. 2017; 11(2): 87–92. https://doi.org/10.5114/hpc.20....
29.
Aguero-Rosenfeld ME, Wang G, Schwarz I, Womser GP. Diagnosis of Lyme borreliosis. Clin Microbiol Rev. 2005; 18(3): 484–509. https://doi.org/10.1128/CMR.18....
30.
Diterich I, Härter L, Hassler D, Wendel A, Hartung T. Modulation of Cytokine Release in Ex Vivo-Stimulated Blood from Borreliosis Patients Infect Immun. 2001; 69(2): 687–694. https://doi.org/10.1128/IAI.69....
31.
Kisand KE, Prükk T, Kissand KV, Lüüs SM, Kalbe I, Uibo R. Propensity to excessive proinflammmatory response in chronic Lyme borreliosis. APMIS. 2007; 115: 134–141. https://doi.org/10.1111/j.1600....
32.
Shin JJ, Strle K, Glickstein LJ, Luster AD, Steere AC. Borrelia burgdorferi stimulation of chemokine secretion by cells of monocyte lineage in patients with Lyme arthritis. Arthritis Res Ther. 2010; 12(5): R168. https://doi.org/10.1186/ar3128.
33.
Liu M, Guo S, Hibbert JM, Jain V, Singh N, Wilson NO, et al. CXCL10/IP-10 in Infectious Diseases Pathogenesis and Potential Therapeutic Implications. Cytokine Growth Factor Rev. 2011; 22(3): 121–130. https://doi.org/10.1016/j.cyto....
34.
Nordberg M, Forsberg P, Johansson A, Nyman D, Jansson C, Ernerudh J, et al. Cytotoxic mechanisms may play a role in the local immune response in the central nervous system in neuroborreliosis. J Neuroimmunol. 2011; 232(1): 186–193. https://doi.org/10.1016/j.jneu....
35.
Sallusto F, Lanzavecchia A. Human Th17 cells in infection and autoimmunity. Microbes Infect. 2009; 11(5): 620–624. https://doi.org/10.1016/j.mici....
36.
Zhi H, Xie J, Skare JT. The Classical Complement Pathway Is Required to Control Borrelia burgdorferi Levels During Experimental Infection. Front Immunol. 2018; 9: 959. https://doi.org/10.3389/fimmu.....
37.
Sjöwall J, Carlsson A, Vaarala O, Bergström S, Ernerudh J, Forsberg PC, et al. Innate immune response in Lyme borreliosis: enhanced tumor necrosis factor-? and interleukin-12 in asymptomatic individuals in response to live spirochetes. Clin Exp Immunol. 2005; 141: 89–98. https://doi.org/10.1111/j.1365....
38.
Jarefors S, Bennet L, You E, Forsberg P, Ekerfelt C, Berglund J, et al. Lyme borreliosis reinfection: might it be explained by a gender difference in immune response? Immunology. 2006; 118: 224–232. https://doi.org/10.1111/j.1365....
39.
Cerar T, Ogrinc K, Lotrič-Furlan S, Kobal J, Levičnik-Stezinar S, Strle F, et al. Diagnostic Value of Cytokines and Chemokines in Lyme Neuroborreliosis. Clin Vaccine Immunol. 2013; 20(10): 1578–1584. https://doi.org/10.1128/CVI.00....
40.
Burgasova OA, Uskov AN, Grichenko NE, Tseneva GIa. Features of cytokine levels in serum of patients with tick-borne borreliosis with different clinical signs. Zh Mikrobiol Epidemiol Immunobiol. 2010; 3: 67–71.
41.
Urbaniak J, Dybaś E, Krajewska M, Weyde W, Klinger M, Woźniak M. The assessment of usefulness of gelatin zymography for metalloproteinase 9 (MMP-9) determination in plasma. Diagn Lab. 2007; 43: 657–667.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.