Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Prevalence and antimicrobial susceptibility of Listeria monocytogenes strains isolated from a
meat processing plant
1
Department of Microbiology and Food Technology, UTP University of Science and Technology, Bydgoszcz, Poland
2
Plant Breeding and Acclimatization Institute – National Research Institute, Bydgoszcz, Poland
3
Institute of Telecommunications and Computer Science, UTP University of Science and Technology, Bydgoszcz, Poland
4
Department of Microbiology, Nicolaus Copernicus University in Toruń, Collegium Medicum of L. Rydygier in Bydgoszcz,
Poland
5
Department of Food Hygiene and Consumer Health, University of Environmental and Life Sciences, Wrocław, Poland
6
Department of Theoretical Foundations of Biomedical Sciences and Medical Computer Science, Nicolaus Copernicus
University, Toruń, L. Rydygier Collegium Medium, Bydgoszcz, Poland
Corresponding author
Krzysztof Skowron
Department of Microbiology, Nicolaus Copernicus University in Toruń, Collegium Medicum of L. Rydygier in Bydgoszcz, M. Skłodowskiej-Curie 9, 85-094, Bydgoszcz, Poland
Department of Microbiology, Nicolaus Copernicus University in Toruń, Collegium Medicum of L. Rydygier in Bydgoszcz, M. Skłodowskiej-Curie 9, 85-094, Bydgoszcz, Poland
Ann Agric Environ Med. 2021;28(4):595-604
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
The ability of L. monocytogenes to create biofilm results in the higher resistance to disinfectants and determines the need to search for effective methods of eradication. The aim of the study was to assess the level of L. monocytogenes contamination in the environment of a meat processing plant. The sensitivity of tested isolates to various antimicrobials used for disinfection purposes was also estimated.
Material and methods:
The samples were taken from raw materials, semi-finished and final products, as well as food contact surfaces inthe production hall and deli meat packaging department. The number of L. monocytogenes and the effect of eight different biocides on bacteria planktonic forms and biofilm formed on stainless steel and polypropylene was investigated. The effect of blood and albumin on L. monocytogenes resistance to disinfectants was also analysed.
Results:
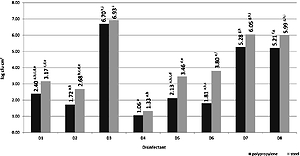
The prevalence of L. monocytogenes on food contact surfaces was estimated at 2.93% (10 of 340 swabs taken). The samples of raw and processed products were not contaminated. Various disinfectants reduced the growth of planktonic L. monocytogenes forms at both tested concentrations 0.5% and 0.1% (irrespective of time exposure). The highest efficacy against L. monocytogenes biofilm was reported for agents containing hydrogen peroxide. The reduction of bacteria number ranged from 6.93–7.21 log CFU × cm-2, and was dependent on the surface type and time of agent application.
Conclusions:
In this study, the effectiveness of various disinfectants against planktonic bacteria and Listeria biofilm was observed. For the majority of disinfectants, the extension of time exposure increased bacteria elimination from the biofilm. The presence of blood resulted in reduction of the antilisterial action of most of the disinfectants applied at low concentrations.
The ability of L. monocytogenes to create biofilm results in the higher resistance to disinfectants and determines the need to search for effective methods of eradication. The aim of the study was to assess the level of L. monocytogenes contamination in the environment of a meat processing plant. The sensitivity of tested isolates to various antimicrobials used for disinfection purposes was also estimated.
Material and methods:
The samples were taken from raw materials, semi-finished and final products, as well as food contact surfaces inthe production hall and deli meat packaging department. The number of L. monocytogenes and the effect of eight different biocides on bacteria planktonic forms and biofilm formed on stainless steel and polypropylene was investigated. The effect of blood and albumin on L. monocytogenes resistance to disinfectants was also analysed.
Results:
The prevalence of L. monocytogenes on food contact surfaces was estimated at 2.93% (10 of 340 swabs taken). The samples of raw and processed products were not contaminated. Various disinfectants reduced the growth of planktonic L. monocytogenes forms at both tested concentrations 0.5% and 0.1% (irrespective of time exposure). The highest efficacy against L. monocytogenes biofilm was reported for agents containing hydrogen peroxide. The reduction of bacteria number ranged from 6.93–7.21 log CFU × cm-2, and was dependent on the surface type and time of agent application.
Conclusions:
In this study, the effectiveness of various disinfectants against planktonic bacteria and Listeria biofilm was observed. For the majority of disinfectants, the extension of time exposure increased bacteria elimination from the biofilm. The presence of blood resulted in reduction of the antilisterial action of most of the disinfectants applied at low concentrations.
REFERENCES (56)
1.
Saraiva C, García Díez J, Fontes M, et al. Modeling the Behavior of Listeria monocytogenes in Meat. 2018; http://dx.doi.org/10.5772/inte....
2.
Shamloo E, Moghadam ZA, Nazari K, et al. Long Term Survival of Listeria monocytogenes in Stress Conditions: High pH and Salt Concentrations. J Res Med Dent Sci. 2018; 6, 96–100.
3.
Gandhi M, Chikindas LM. Listeria: A foodborne pathogen that knows how to survive. Int J Food Microbiol. 2007; 113, 1–15.
4.
European Union One Health. (2018). Zoonoses Report. (2019). https://www.efsa.europa.eu/en/... Accessed 15 February 2020.
5.
Health Department of Republic of South Africa. (2018). Listeriosis outbreak situation report – 27/04/2018. WHO Country Emergency Preparedness and Readiness Technical Meeting Listeriosis Outbreak in collaboration with MoH and Partners. Johannesburg 19–21 April 2018.
6.
ECDC. (2018). A long term (2015–2018) multi-country outbreak of L. monocytogenes ST6 resulted in 47 confirmed listeriosis cases and 10 fatalities.
8.
Gómez D, Iguácel LP, Rota MC, et al. Occurrence of Listeria monocytogenes in Ready-to-Eat Meat Products and Meat Processing Plants in Spain. Foods, 2015; 14, 271–282. https://doi.org/10.3390/foods4....
9.
Dade-Robertson M, Keren-Paz A, Zhang M, et al. Architects of nature: growing buildings with bacterial biofilms. Microb Biotechnol. 2017; 10, 1157–1163. https://doi.org/10.1111/1751-7....
10.
Bridier A, Sanchez-Vizuete P, Guilbaud M, et al. Biofilm-associated persistence of food-borne pathogens. Food Microbiol. 2015; 45: 167–178. doi: 10.1016/j.fm.2014.04.015.
11.
Baranowska M, Chojnowski W, Nowak H. Disinfection in diary plants. Nauki Inżynieryjskie i Technologie. 2014; 4(15): 9–22.
12.
Coughlan LM, Cotter PD, Hill C, et al. New weapons to fight old enemies: novel strategies for the (bio)control of bacterial biofilms in the food industry. Front Microbiol. 2016; 7: 1641.
13.
El-Azizi M, Farag N, Khardori N. Efficacy of selected biocides in the decontamination of common nosocomial bacterial pathogens in biofilm and planktonic forms. Comp Immunol Microbiol Infect Dis. 2016; 47: 60–71.
14.
Van Houdt R, Michiels CW. Biofilm formation and the food industry, a focus on the bacterial outer surface. J Appl Microbiol. 2010; 109(4): 1117–1131. doi: 10.1111/j.1365-2672.2010.04756.x.
15.
Li J, Feng J, Ma L, et al. Effects of meat juice on biofilm formation of Campylobacter and Salmonella. Int J Food Microbiol. 2017; 253: 20–28. https://doi.org/10.1016/j.ijfo....
16.
Fagerlund A, Moretro T, Heir E, et al. Cleaning and Disinfection of Biofilms Composed of Listeria monocytogenes and Background Microbiota from Meat Processing Surfaces. Appl Environ Microbiol. 2017; 83(17). doi: 10.1128/AEM.01046-17.
17.
Doijad SP, Barbuddhe SB, Garg S, et al. Biofilm-Forming Abilities of Listeria monocytogenes Serotypes Isolated from Different Sources. PLoS ONE. 2015; 10(9). https://doi.org/10.1371/journa....
18.
PN-ISO 18593: 2004. Microbiology of Food and Animal Feeding Stuffs—Horizontal Methods for Sampling Techniques from Surfaces Using Contact Plates and Swabs (ISO 18593:2004) Polish Committee for Standardization.
19.
PN-ISO 11290-1: 2017. Microbiology of the food chain — Horizontal method for the detection and enumeration of Listeria monocytogenes and of Listeria spp. — Part 1: Detection method (ISO 11290-1:2017) Polish Committee for Standardization.
20.
Leclercq A, Clermont D, Bizet C, et al. Listeria rocourtiae sp. nov. Int J Syst Evol Microbiol. 2010; 60, 2210–2214. https://doi.org/10.1099/ijs.0.....
21.
Skowron K, Hulisz K, Gryń G, et al. Comparison of selected disinfectants efficiency against Listeria monocytogenes biofilm formed on various surfaces. Int Microbiol. 2018; 21: 23–33.
22.
Park S, Jung J, Choi S, et al. Molecular Characterization of Listeria monocytogenes Based on the PFGE and RAPD in Korea. Adv Microbiol. 2012; 2: 605–616. doi: 10.4236/aim.2012.24079.
23.
Ozbey G, Hasan Ertas HB, Filizkok F. Prevalence of Listeria species in camel sausages from retail markers in Aydin province in Turkey and RAPD analysis of Listeria monocytogenes isolates. Irish Vet J. 2006; 59: 342–344.
24.
Krzywicka H, Janowska J, Tadeusiak B, et al. Metoda określania stężeń użytkowych preparatów dezynfekcyjnych. Metoda nośnikowa. Warszawa: Wydawnictwo Metodyczne Państwowego Zakładu Higieny; 1993.
25.
PN-ISO 1276:2000. Chemiczne środki dezynfekcyjne i antyseptyczne -- Ilościowa zawiesinowa metoda określania działania bakteriobójczego chemicznych środków dezynfekcyjnych i antyseptycznych stosowanych w sektorze żywnościowym, warunkach przemysłowych i domowych oraz zakładach użyteczności publicznej – Metoda badania i wymagania (faza 2, etap 1).
26.
The Brussel Times: Five dead in listeria outbreaks in the Netherlands and Germany 2019. https://www.brusselstimes.com/.... Accessed 15 February 2020.
27.
Muhterem-Uyar M, Dalmasso M, Bolocan A, et al. Environmental sampling for Listeria monocytogenes control in food processing facilities reveals three contamination scenarios. Food Control. 2015; 51: 94–107. https://doi.org/10.1016/j.food....
28.
Zuber I, Lakicevic B, Pietzka A, et al. Molecular characterization of Listeria monocytogenes isolates from a small-scale meat processor in Montenegro, 2011–2014. Food Microbiol. 2019; 79: 116–122. https://doi.org/10.1016/j.fm.2....
29.
Skowron K, Wałecka-Zacharska E, Wiktorczyk-Kapischke N, et al. Assessment of the Prevalence and Drug Susceptibility of Listeria monocytogenes Strains Isolated from Various Types of Meat. Foods. 2020; 9: 1293. https://doi.org/10.3390/foods9....
30.
Martin B, Perich A, Gómez D, et al. Diversity and distribution of Listeria monocytogenes in meat processing plants. Food Microbiol. 2014; 44: 119–27. https://doi.org/10.1016/j.fm.2....
31.
Buchanan RL, Gorris LGM, Hayman MM, et al. A review of Listeria monocytogenes: An update on outbreaks, virulence, dose-response, ecology, and risk assessments. Food Control 2017; 75: 1–13. https://doi.org/10.1016/j.food....
32.
Skowron K, Wiktorczyk N, Grudlewska K, et al. Drug-susceptibility, biofilm-forming ability and biofilm survival on stainless steel of Listeria spp. strains isolated from cheese. Int J Food Microbiol. 2019; 2: 75–82. htps://doi.org/10.1016/j.ijfoodmicro.2019.02.021.
33.
Cabeça T, Pizzolitto A, Pizzolitto E. Activity of disinfectants against foodborne pathogens in suspension and adhered to stainless steel surfaces. Braz J Microbiol. 2012; 43: 1112–1119. http://dx.doi.org/10.1590/S151....
34.
Aarnisalo K, Salo S, Miettinen H, Suihko ML, et al. Bactericidal efficiencies of commercial disinfectants against Listeria monocytogenes on surfaces. J Food Saf. 2000; 20: 237–250. http://doi: 10.1111/j.1745-4565.2000.tb00302.x.
35.
Addeen A, Benjakul S, Maqsood S, et al. Chicken blood promotes growth of Listeria monocytogenes, Salmonella Typhimurium, Campylobacter jejuni and Pseudomonas aeruginosa in minced chicken during refrigerated storage. Int Food Res J. 2015; 22: 2619–2728.
36.
Wiktorczyk N, Grudlewska K, Skowron K, et al. The effect of blood on the ability of biofilm formation by Listeria monocytogenes strains. Med Res J. 2018; 3, 28–31. doi: 10.5603/MRJ.2018.0005.
37.
Skowron K, Wałecka-Zacharska E, Grudlewska K, et al. Disinfectant Susceptibility of Biofilm Formed by Listeria monocytogenes under Selected Environmental Conditions. Microorganisms. 2019; 7: 280. doi: 10.3390/microorganisms7090280.
38.
Kyoui D, Hirokawa E, Takahashi H, et al. Effect of glucose on Listeria monocytogenes biofilm formation, and assessment of the biofilm’s sanitation tolerance. Biofouling. 2016; 32: 815–826. doi: 10.1080/08927014.2016.1198953.
39.
Yun HS, Kim Y, Oh S, et al. Susceptibility of Listeria monocytogenes biofilms and planktonic cultures to hydrogen peroxide in food processing environments. Biosci Biotechnol Biochem. 2012; 76: 2008–2013. https://doi.org/10.1271/bbb.12....
40.
Pang X, Yuk HG. Effects of the colonization sequence of Listeria monocytogenes and Pseudomonas fluorescens on survival of biofilm cells under food-related stresses and transfer to salmon. Food Microbiol. 2019; 82: 142–150. https://doi.org/10.1016/j.fm.2....
41.
Rodríguez-Melcón C, Riesco-Peláez F, García-Fernández C, et al. Susceptibility of Listeria monocytogenes planktonic cultures and biofilms to sodium hypochlorite and benzalkonium chloride. Food Microbiol. 2019; 82: 533–540. g/10.1016/j.fm.2019.03.020.
42.
Smith K, Hunter IS. Efficacy of common hospital biocides with biofilms of multi-drug resistant clinical isolates. J. Med. Microbiol. 2008; 57: 966–973. doi: 10.1099/jmm.0.47668-0.
43.
Mah TF, O’Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001; 9: 34–39. https://doi.org/10.1016/S0966-....
44.
Hou J, Wang C, Rozenbaum RT, Gusnaniar N, et al. Bacterial Density and Biofilm Structure Determined by Optical Coherence Tomography. Sci Rep. 2019; 9: 9794. https://doi.org/10.1038/s41598....
45.
Barbosa J, Grzybowski V, Cuppini M, et al. Listeria monocytogenes adhesion to food processing surfaces (boning knives) and the removal efficacy of different sanitizers. Ital J Food Sci. 2016; 28: 733–743. https://doi.org/10.14674/1120-....
46.
Bas S, Kramer M, Stopar D. Biofilm Surface Density Determines Biocide Effectiveness. Front Microbiol. 2017; 8: 2443. doi: 10.3389/fmicb.2017.02443.
47.
Reis-Teixeira FBD, Alves VF, Martinis ECP. Growth, viability and architecture of biofilms of Listeria monocytogenes formed on abiotic surfaces. Braz J Microbiol. 2017; 48: 587–591. https://doi.org/10.1016/j.bjm.....
48.
Russo P, Hadjilouka A, Beneduce L, et al. Effect of different conditions on Listeria monocytogenes biofilm formation and removal. Czech J Food Sci. 2018; 36: 208–214. https://doi.org/10.17221/199/2....
49.
Poimenidou SV, Chrysadaakou M, Tzakoniati A, et al. Variability of Listeria monocytogenes strains in biofilm formation on stainless steel and polystyrene materials and resistance to peracetic acid and quaternary ammonium compounds. Int J Food Microbiol. 2017; 237: 164–171.
50.
Skowron K, Grudlewska K, Krawczyk A, et al. The effectiveness of radiant catalytic ionization in inactivation of Listeria monocytogenes planktonic and biofilm cells from food and food contact surfaces as a method of food preservation. J App Microbiol. 2018; 124: 1493–1505. https://doi.org/10.1111/jam.13....
51.
Lineback CB, Nkemngong CA, Wu ST, et al. Hydrogen peroxide and sodium hypochlorite disinfectants are more effective against Staphylococcus aureus and Pseudomonas aeruginosa biofilms thanquaternary ammonium compounds. Antimicrob Resist Infect Control, 2018; 17: 154. https://doi.org/10.1186/s13756....
52.
Zineb G, Mliji M, Youssef G, et al. Anti-Adhesion and Anti-Biofilm Effectiveness of Disinfectants Used In Hemodialysis against both Staphylococcus Warneri and Staphylococcus Sciuri Biofilms. Int J Eng Res App. 2014; 4: 86–96.
53.
Boyce MJ, Guercia KA, Sullivan L, et al. Prospective cluster controlled crossover trial to compare the impact of an improved hydrogen peroxide disinfectant and a quaternary ammonium based disinfectant on surface contamination and health care outcomes. Am J Infect Control. 2017; 45: 1006–1010. https://doi.org/10.1016/j.ajic....
54.
Best M, Kennedy ME, Coates F. Efficacy of a Variety of Disinfectants against Listeria spp. Appl Environ Microbiol. 1990; 56(2): 377–380.
55.
Overney A, Jacques-André-Coquin J, Ng P, et al. Impact of environmental factors on the culturability and viability of Listeria monocytogenes under conditions encountered in food processing plants. Int J Food Microbiol. 2017; 244: 74–81. https://doi.org/10.1016/j.ijfo....
56.
Ren TJ, Frank JF. Susceptibility of Starved Planktonic and Biofilm Listeria monocytogenes to Quaternary Ammonium Sanitizer as Determined by Direct Viable and Agar Plate Counts. J Food Prot. 1993; 56: 573–576. doi: 10.4315/0362-028X-56.7.573.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.