Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Is the needle still needed? Relationship between laboratory features of pleural effusion and results of lung CT examinations in patients with pneumonia and lung cancer
1
Chair and Department of Pneumonology, Oncology and Allergology, Medical University, Lublin, Poland
2
Department of Radiology and Nuclear Medicine, Independent Public Clinical Hospital No. 4, Lublin, Poland
3
Department of Cardiology, Independent Public Provincial Specialist Hospital, Chełm, Poland
4
Department of Pharmaceutical Botany, Medical University, Lublin, Poland
Corresponding author
Anna Grzywa-Celińska
Chair and Department of Pneumonology, Oncology and Allergology, Medical University, Lublin, Poland
Chair and Department of Pneumonology, Oncology and Allergology, Medical University, Lublin, Poland
Ann Agric Environ Med. 2023;30(2):224-228
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Thoracocentesis is an invasive procedure routinely performed in the diagnosis of causes for the presence of pathological fluid in the pleural cavity. In many patients, a computed tomography scanning (CT) is also performed to diagnose the cause of the presence of fluid in the pleural cavity. The diagnostic value of CT is particularly high in situations in which performing thoracocenthesis could be associated with an increased risk of complications. The aim other study was to assess the relationship between the objective radiological features and the results of laboratory tests of fluid collected by thoracocenthesis in patients with pneumo-nias (n=18) and lung cancer (n=35).
Material and methods:
The examined group consisted of the patients with pneumonia (n=18) and lung cancer (n=35) which resulted in the presence of fluid in the pleural cavity. In the patients thoracocentesis, CT lung scanning was also performed, according to the medical indications. Three scans with the greatest amount of fluid were identified, and the mean density of the fluid expressed in Hounsfield units was calculated within the area. These calculations were compared with the results of laboratory fluid tests.
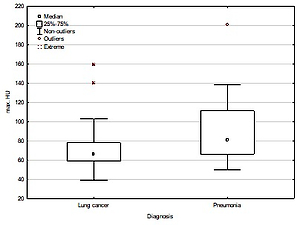
Results:
The maximum number of Hounsfield units (HU) was significantly lower in the group of lung cancer patients, compared to those diagnosed with pneumonia (74.3% sensi-tivity and 55.6% specificity). The pH of pleural fluid was significantly lower in patients with lung cancer, compared to those with pneumonia (74.3% sensitivity and 66.7% specificity).
Conclusions:
According to the results, radiological differentiation of pneumonia and lung cancer resulting in pleural effusion, to some extent is possible; however, the needle is still needed.
Thoracocentesis is an invasive procedure routinely performed in the diagnosis of causes for the presence of pathological fluid in the pleural cavity. In many patients, a computed tomography scanning (CT) is also performed to diagnose the cause of the presence of fluid in the pleural cavity. The diagnostic value of CT is particularly high in situations in which performing thoracocenthesis could be associated with an increased risk of complications. The aim other study was to assess the relationship between the objective radiological features and the results of laboratory tests of fluid collected by thoracocenthesis in patients with pneumo-nias (n=18) and lung cancer (n=35).
Material and methods:
The examined group consisted of the patients with pneumonia (n=18) and lung cancer (n=35) which resulted in the presence of fluid in the pleural cavity. In the patients thoracocentesis, CT lung scanning was also performed, according to the medical indications. Three scans with the greatest amount of fluid were identified, and the mean density of the fluid expressed in Hounsfield units was calculated within the area. These calculations were compared with the results of laboratory fluid tests.
Results:
The maximum number of Hounsfield units (HU) was significantly lower in the group of lung cancer patients, compared to those diagnosed with pneumonia (74.3% sensi-tivity and 55.6% specificity). The pH of pleural fluid was significantly lower in patients with lung cancer, compared to those with pneumonia (74.3% sensitivity and 66.7% specificity).
Conclusions:
According to the results, radiological differentiation of pneumonia and lung cancer resulting in pleural effusion, to some extent is possible; however, the needle is still needed.
REFERENCES (14)
1.
Feller-Kopman D, Light R. Pleural Disease. N Engl J Med. 2018;378:740–751. https://doi.org/10.1056/NEJMra....
2.
Massie J. My love affair with the pleural space. Med J Australia. 2020;213:526–528.e1. https://doi.org/10.5694/mja2.5....
3.
Burstiner L, Al Khalili Y. Anatomy, Thorax, Pleurae. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing, 2021.
4.
Dhanasopon AP, Blasberg JD, Mase VJ Jr. Surgical Management of Pneumothorax and Pleural Space Disease. Surg Clin North Am. 2022;102 (3):413–427. https://doi.org/10.1016/j.suc.....
5.
Porcel JM, Light RW. Pleural Fluid Analysis: Are Light’s Criteria Still Relevant After Half a Century? Clin Chest Med. 2021;42(4):599–609. https://doi.org/10.1016/j.ccm.....
6.
Beaudoin S, Gonzalez AV. Evaluation of the patient with pleural effusion. CMAJ. 2018;190(10): E291-E295.https://doi.org/10.1503/cmaj.1....
7.
Hayama N, Hattori S, Takahashi G, et al. Cytokine/Chemokine/Growth Factor Levels in Malignant Pleural Effusion of Non-small Cell Lung Cancer. Tokai J Exp Clin Med. 2020;45(4): 224–229.
8.
Bassen DM, Nayak S, Li X, et al. Clinical Decision Support System (CDSS) for the Classification of Atypical Cells in Pleural Effusions. Complex Adaptive Systems. 2013;20:379–384.
9.
Rodríguez-Panadero F, López Mejías J. Low glucose and pH levels in malignant pleural effusions. Diagnostic significance and prognostic value in respect to pleurodesis. Am Rev Respir Dis. 1989;139(3):663–7.
10.
Addala DN, Bedawi EO, Rahman NM. Parapneumonic Effusion and Empyema. Clin Chest Med. 2021;42(4):637–647. https://doi.org/10.1016/j.ccm.....
11.
Good JT Jr, Taryle DA, Maulitz RM. The diagnostic value of pleural fluid pH. Chest. 1980;78(1):55–9.
12.
Yalçin-Şafak K, Umarusman-Tanju N, et al. Efficacy of Computed Tomography (CT) Attenuation Values and CT Findings in the Differentiation of Pleural Effusion. Pol J Radiol. 2017;82:100–105. https://doi.org/10.12659/PJR.8....
13.
Porcel JM, Pardina M, Bielsa S, et al. Derivation and validation of a CT scan scoring system for discriminating malignant from benign pleural effusions. Chest. 2015;147(2):513–519. https://doi.org/10.1378/chest.....
14.
Lee MS, Oh JY, Kang CI, et al. Guideline for Antibiotic Use in Adults with Community-acquired Pneumonia. Infect Chemother. 2018;50(2):160–198. https://doi.org/10.3947/ic.201....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.