Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Antibiofilm activity of selected plant essential oils from the Lamiaceae family against Candida albicans clinical isolates
1
University of Veterinary Medicine and Pharmacy, Košice, Slovak Republic
Corresponding author
Martina Proškovcová
The University of Veterinary Medicine and Pharmacy in Košice, Komenského, 04181, Košice, Slovak Republic
The University of Veterinary Medicine and Pharmacy in Košice, Komenského, 04181, Košice, Slovak Republic
Ann Agric Environ Med. 2021;28(2):260-266
KEYWORDS
TOPICS
- Biological agents posing occupational risk in agriculture, forestry, food industry and wood industry and diseases caused by these agents (zoonoses, allergic and immunotoxic diseases)
- State of the health of rural communities depending on various factors: social factors, accessibility of medical care, etc.
ABSTRACT
Introduction:
The virulence of Candida albicans is conditioned by several virulence factors, one of which is the formation of biofilm which reduces the sensitivity of the yeast to conventional antimycotics. This study determines the antifungal and antibiofilm activity of five essential oils (EOs) of the Lamiaceae family: Salvia officinalis, Thymus vulgaris, Rosmarinus officinalis, Origanum vulgare, and Hyssopus officinalis.
Material and methods:
In the preliminary research, the antifungal effect of eachof the EOs was tested in the concentration range of 200–0.4 mg/mL on planktonic Candida albicans (C. albicans) cells. A total of 13 C. albicans clinical isolates and one reference strain were evaluated on biofilm formation.
Results:
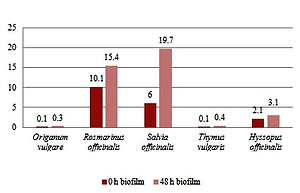
Nine isolates (69.2%) showed weak biofilm production and four strains (30.8%) were detected as moderate biofilm producers. The EOs of Thymus vulgaris and Origanum vulgare were seen as effective antifungal agents on planktonic cells with the MIC 0.4 mg/mL. The highest average MIC values were recorded in Salvia officinalis EO (24.0 and 14.8 mg/mL). All isolates were used to determine EOs efficacy on the inhibition of adherence phase and biofilm formation. The biofilm production of C. albicans after exposition by EOs was quantitatively examined by crystal violet dye.
Conclusions:
The most effective for adherence phase and biofilm formation were EOs of Origanum vulgare (0.1 mg/mL and 0.3 mg/mL) and Thymus vulgaris (0.1 mg/mL and 0.4 mg/mL). The obtained results show that EOs of Thymus vulgaris and Origanum vulgare are potential agents for antifungal treatment or prophylaxis by reducing the resistance of pathogen.
The virulence of Candida albicans is conditioned by several virulence factors, one of which is the formation of biofilm which reduces the sensitivity of the yeast to conventional antimycotics. This study determines the antifungal and antibiofilm activity of five essential oils (EOs) of the Lamiaceae family: Salvia officinalis, Thymus vulgaris, Rosmarinus officinalis, Origanum vulgare, and Hyssopus officinalis.
Material and methods:
In the preliminary research, the antifungal effect of eachof the EOs was tested in the concentration range of 200–0.4 mg/mL on planktonic Candida albicans (C. albicans) cells. A total of 13 C. albicans clinical isolates and one reference strain were evaluated on biofilm formation.
Results:
Nine isolates (69.2%) showed weak biofilm production and four strains (30.8%) were detected as moderate biofilm producers. The EOs of Thymus vulgaris and Origanum vulgare were seen as effective antifungal agents on planktonic cells with the MIC 0.4 mg/mL. The highest average MIC values were recorded in Salvia officinalis EO (24.0 and 14.8 mg/mL). All isolates were used to determine EOs efficacy on the inhibition of adherence phase and biofilm formation. The biofilm production of C. albicans after exposition by EOs was quantitatively examined by crystal violet dye.
Conclusions:
The most effective for adherence phase and biofilm formation were EOs of Origanum vulgare (0.1 mg/mL and 0.3 mg/mL) and Thymus vulgaris (0.1 mg/mL and 0.4 mg/mL). The obtained results show that EOs of Thymus vulgaris and Origanum vulgare are potential agents for antifungal treatment or prophylaxis by reducing the resistance of pathogen.
FUNDING
Funding source
This research was financial supported by IGA UVLF 05/2020 „In vitro“ determination of proapoptotic, antibiofilm and antioxidant activity of selected essential oils from plants of the Lamiaceae family“ and by the Slovak Research and Development Agency under the contract No. APVV-15-0377.
REFERENCES (45)
1.
Martins N, Ferreira IC, Barros L, et al. Candidiasis: predisposing factors, prevention, diagnosis and alternative treatment. Mycopathologia. 2014; 177(5–6): 223–240. https://doi.org/10.1007/s11046....
2.
Moore D, Alexopoulos CJ, Ahmadjian V. Fungus, Encyclopadia Britannica, Encyclopadia Britannica, inc, (access 2020.04.05). https://www.britannica.com/sci....
3.
Khan HA, Baig FK, Mehboob R. Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pac J Trop Biomed. 2017; 7(5): 478–82. https://doi.org/10.1016/j.apjt....
4.
Gow NA, Yadav B. Microbe Profile: Candida albicans: a shape-changing, opportunistic pathogenic fungus of humans. Microbiology. 2017; 163(8): 1145–1147. https://doi.org/10.1099/mic.0.....
5.
D’agostino M, Tesse N, Frippiat JP, et al. Essential oils and their natural active compounds presenting antifungal properties. Molecules. 2019 Jan; 24(20): 3713. https://doi.org/10.3390/molecu....
6.
Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence. 2013; 4(2): 119–128. https://doi.org/10.4161/viru.2....
7.
Fanning S, Mitchell AP. Fungal biofilms. PLoS Pathog. 2012; 8(4): e1002585. https://doi.org/10.1371/journa....
8.
Lohse MB, Gulati M, Johnson AD, et al. Development and regulation of single-and multi-species Candida albicans biofilms. Nat Rev Microbiol. 2018; 16(1): 19–31. https://doi.org/10.1038/nrmicr....
9.
Höfs S, Mogavero S, Hube B. Interaction of Candida albicans with host cells: virulence factors, host defense, escape strategies, and the microbiota. J Microbiol. 2016; 54(3): 149–169. http://doi.org/10.1007/s12275-....
10.
Tsui C, Kong EF, Jabra-Rizk MA. Pathogenesis of Candida albicans biofilm. FEMS Pathog Dis. 2016; 74(4): ftw018. https://doi.org/10.1093/femspd....
11.
Pristov KE, Ghannoum MA. Resistance of Candida to azoles and echinocandins worldwide. Clin Microbiol Infect. 2019; 25(7): 792–8. https://doi.org/10.1016/j.cmi.....
12.
Costa-de-Oliveira S, Rodrigues AG. Candida albicans Antifungal Resistance and Tolerance in Bloodstream Infections: The Triad Yeast-Host-Antifungal. Microorganisms. 2020; 8(2): 154. https://doi.org/10.3390/microo....
13.
Raja RR. Medicinally potential plants of Labiatae (Lamiaceae) family: an overview. Res J Med Plant. 2012; 6(3): 203–213. https://doi.org./10.3923/rjmp.....
14.
Carović-Stanko K, Petek M, Martina G, et al. Medicinal plants of the family Lamiaceaeas functional foods–a review. Czech J Food Sci. 2016; 34(5): 377. https://doi.org/10.17221/504/2....
15.
Nieto G. Biological activities of three essential oils of the Lamiaceae family. Medicines. 2017; 4(3): 63. https://doi.org/10.3390/medici....
16.
Nagy M, Mučaji P, Grančai D. Pharmacognosy. Biologically active plant metabolites and their sources. 2nd ed. Bratislava: HERBA, 2017. ISBN 978-80-89631-64-3.
17.
Sookto T, Srithavaj T, Thaweboon S et al. In vitro effects of Salvia officinalis L. essential oil on Candida albicans. Asian Pac J Trop Biomed. 2013; 3(5): 376–380. https://doi.org/10.1016/S2221-....
18.
El Euch SK, Hassine DB, Cazaux S, et al. Salvia officinalis essential oil: Chemical analysis and evaluation of anti-enzymatic and antioxidant bioactivities. S Afr J Bot. 2019; 120: 253–60. https://doi.org/10.1016/j.sajb....
19.
Sakkas H, Papadopoulou C. Antimicrobial activity of basil, oregano, and thyme essential oils. J Microbiol Biotechnol. 2017; 27(3): 429–38. https://doi.org/10.4014/jmb.16....
20.
Gucwa K, Milewski S, Dymerski T, et al. Investigation of the antifungal activity and mode of action of Thymus vulgaris, Citrus limonum, Pelargonium graveolens, Cinnamomum cassia, Ocimum basilicum, and Eugenia caryophyllus essential oils. Molecules. 2018; 23(5): 1116. https://doi.org/10.3390/molecu....
21.
Elansary HO, Abdelgaleil SA, Mahmoud EA, et al. Effective antioxidant, antimicrobial and anticancer activities of essential oils of horticultural aromatic crops in northern Egypt. BMC Complement Altern Med. 2018; 18(1): 214. https://doi.org/10.1186/s12906....
22.
Alizadeh BB, Shahidi F. Melissa officinalis essential oil: Chemical compositions, antioxidant potential, total phenolic content and antimicrobial activity. Nutr Food Sci Res. 2019; 6(1): 17–25. https://doi.org/10.29252/nfsr.....
23.
Serra E, Saubade F, Ligorio C, et al. Methylcellulose hydrogel with Melissa officinalis essential oil as a potential treatment for oral candidiasis. Microorganisms. 2020; 8(2): 215. https://doi.org/10.3390/microo....
24.
Desam NR, Al-Rajab AJ, Sharma M, et al. Chemical constituents, in vitro antibacterial and antifungal activity of Mentha× Piperita L. (peppermint) essential oils. J King Saud Univ Sci. 2019; 31(4): 528–33. https://doi.org/10.1016/j.jksu....
25.
Modarresi M, Farahpour MR, Baradaran B. Topical application of Mentha piperita essential oil accelerates wound healing in infected mice model. Inflammopharmacol. 2019; 27(3): 531–7. https://doi.org/10.1007/s10787....
26.
Raut JS, Karuppayil SM. A status review on the medicinal properties of essential oils. Ind Crops Prod. 2014; 62: 250–264. https://doi.org/10.1016/j.indc....
27.
Jin YYHK, Yip HK, Samaranayake YH, et al. Biofilm-forming ability of Candida albicans is unlikely to contribute to high levels of oral yeast carriage in cases of human immunodeficiency virus infection. J Clin Microbiol. 2003; 41(7): 2961–2967. https://doi.org/10.1128/JCM.41....
28.
Dhale RP, Ghorpade MV, Dharmadhikari CA. Comparison of various methods used to detect biofilm production of Candida species. J Clin Diagn Res: JCDR. 2014; 8(11): DC18-Dc20. http://doi.org/10.7860/JCDR/20....
29.
Ruchi T, Sujata B, Anuradha D. Comparison of phenotypic methods for the detection of biofilm production in uro-pathogens in a tertiary care hospital in India. Int J Curr Microbiol App Sci. 2015; 4(9): 840–49.
30.
Chemsa AE, Zellagui A, Öztürk EE, et al. Antibiofilm formation, antioxidant and anticholinesterase activities of essential oil and methanol extract of Marrubium deserti de Noé. J Mater Environ Sci. 2016; 7(3): 993–1000.
31.
Invasive Candidiasis Statistics. Centers for Disease Control and Prevention. https://www.cdc.gov/fungal/dis... (access 07.08.2020).
32.
Gulati M, Nobile CJ. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016; 18(5): 310–321. http://www.doi.org/10.1016/j.m....
33.
Silva S, Rodrigues CF, Araújo D, et al. Candida species biofilms’ antifungal resistance. J Fungi. 2017; 3(1): 8. https://doi.org/10.3390/jof301....
34.
Bekut M, Brkić S, Kladar N, et al. Potential of selected Lamiaceae plants in anti (retro) viral therapy. Pharmacol Res. 2018; 133: 301–314. https://doi.org/10.1016/j.phrs....
35.
Baj T, Biernasiuk A, Wróbel R, et al. Chemical composition and in vitro activity of Origanum vulgare L., Satureja hortensis L., Thymus serpyllum L. and Thymus vulgaris L. essential oils towards oral isolates of Candida albicans and Candida glabrata. Open Chem. 2020; 18(1): 108–118. https://doi.org/10.1515/chem-2....
36.
Mohandas V, Ballal M. Distribution of Candida species in different clinical samples and their virulence: biofilm formation, proteinase and phospholipase production: a study on hospitalized patients in southern India. J Glob Infect Dis. 2011; 3(1): 4. https://doi.org/10.4103/0974-7....
37.
Udayalaxmi SJ, D’Souza D. Comparison between virulence factors of Candida albicans and non-albicans species of Candida isolated from genitourinary tract. J Clin Diagn Res. 2014; 8(11): DC15-DC17. http://doi.org/10.7860/JCDR/20....
38.
Marak MB, Dhanashree B. Antifungal susceptibility and biofilm production of Candida spp. isolated from clinical samples. Int J Microbiol. 2018; 2018. https://doi.org/10.1155/2018/7....
39.
Sida H, Shah P, Pethani J, et al. Study of biofilm formation as a virulence marker in Candida species isolated from various clinical specimens. Int J Med Sci Public Health. 2016; 5(5): 842–846. https://doi.org/10.5455/ijmsph....
40.
Doke SK, Raut JS, Dhawale S, et al. Sensitization of Candida albicans biofilms to fluconazole by terpenoids of plant origin. J Gen Appl Microbiol. 2014; 60(5): 163–168. https://doi.org/10.2323/jgam.6....
41.
Vasconcelos LCD, Sampaio FC, Albuquerque ADJDR, et al. Cell viability of Candida albicans against the antifungal activity of thymol. Braz Dent J. 2014; 25(4): 277–281. https://doi.org/10.1590/0103-6....
42.
Cavalcanti Y, Almeida LDFDD, Padilha WWN. Anti-adherent activity of Rosmarinus officinalis essential oil on Candida albicans: an SEM analysis. Rev Odonto Cienc. 2011; 26(2): 139–144. http://dx.doi.org/10.1590/S198....
43.
Thaweboon B, Thaweboon S. Inhibitory effect of Salvia officinalis L. oil on Candida biofilm. In Advanced Materials, Mechanical and Structural Engineering: Proceedings of the 2nd International Conference of Advanced Materials, Mechanical and Structural Engineering (AMMSE 2015), Je-ju Island, South Korea, September 18–20, 2015. 2016 Apr 14 (p. 1). CRC Press.
44.
Potente G, Bonvicini F, Gentilomi GA, et al. Anti-Candida Activity of Essential Oils from Lamiaceae Plants from the Mediterranean Area and the Middle East. Antibiotics. 2020; 9(7): 395. https://doi.org/10.3390/antibi....
45.
Suntres ZE, Coccimiglio J, Alipour M. The bioactivity and toxicological actions of carvacrol. Crit Rev Food Sci Nutr. 2015; 55(3): 304–18. http://dx.doi.org/10.1080/1040....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.