Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
CASE REPORT
Meningoencephalitis and postinflammatory hydrocephalus in the course of COVID-19 disease in newborn – the potential role of acetazolamide as add-on therapy to the standard treatment
1
Department of Neonatology and Neonatal Intensive Care Unit, Independent Public Healthcare, Puławy, Poland
2
Department of Veterinary Hygiene, Voivodship Veterinary Inspectorate, Lublin, Poland
Corresponding author
Sławomir Jan Wątroba
Department of Neonatology and Neonatal Intensive Care Unit, Independent Public Healthcare, Bema 1, 24-100 Puławy, Poland
Department of Neonatology and Neonatal Intensive Care Unit, Independent Public Healthcare, Bema 1, 24-100 Puławy, Poland
Ann Agric Environ Med. 2022;29(4):595-602
KEYWORDS
TOPICS
- Biological agents posing occupational risk in agriculture, forestry, food industry and wood industry and diseases caused by these agents (zoonoses, allergic and immunotoxic diseases)
- State of the health of rural communities depending on various factors: social factors, accessibility of medical care, etc.
ABSTRACT
The topic of SARS-CoV-2 coronavirus infections in children is still complex and not fully understood. Acute meningoencephalitis
(ME) was not considered a common presentation of COVID-19 in paediatrics, however, over time, several paediatric patients
with ME associated with SARS-CoV-2 coronavirus infection have been described. The case report describes the clinical case
of a newborn admitted to the Neonatal Intensive Care Unit (NICU) on 11th day of life due to severe SARS-CoV-2 coronavirus
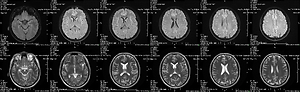
infection, who experienced multiple seizure episodes. The patient was diagnosed with ME and hydrocephalus. In the absence
of clinical improvement, despite the use of standard treatment, acetazolamide (ACZ) was used, achieving complete relief of
seizures and gradual regression of hydrocephalus. This means that ACZ can be considered as an add-on therapy to standard
treatment in cases of ME and postinflammatory hydrocephalus in the course of COVID-19 disease.
ABBREVIATIONS
ABB – acid-base balance; ACV – aciclovir; ACZ – acetazolamide; ALP – alkaline phosphatase; AMP – ampicillin; ARDS – acute
respiratory distress syndrome; CA – carbonate dehydratase; CMV – cytomagalovirus; CNS – central nervous system; CPE –
carbapenamase-producing Enterobacteriaceae; CSF – cerebrospinal fluid; CTX – cefotaxime; ECG – electrocardiogram; EEG
– electroencephalogram; FFP – fresh frozen plasma; FiO2 – oxygen concentration in the breathing mixture; FLC – fluconazole;
FR – ferritin; GBS – group B Streptococcus; GM – gentamicin; HFNC – high-flow nasal cannula; IVH – intraventricular
haemorrhage; LP – lumbar puncture; ME – meningoencephalitis; MP – meropenem; MRI – magnetic resonance imaging;
MRSA – methicillin-resistant Staphylococcus aureus; MSSA – methicillin-sensitive Staphylococcus aureus; NICU – neonatal
intensive care unit; PB – phenobarbital; PC – platelet concentrate; PIMS – paediatric inflammatory multisystem syndrome;
PVH – periventricular haemorrhage; RAV – right axillary vein; RSV – respiratory syncytial virus; RT-PCR – real-time reverse
transcription polymerase chain reaction; SI – singal intensity; TnT – troponin T; USG – ultrasonography; VAN – vancomycin
REFERENCES (27)
1.
Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382(13):1199–1207. http://doi.org/10.1056/NEJMoa2....
2.
Xu B, Gutierrez B, Mekaru S, et al. Epidemiological data from the COVID-19 outbreak, real-time case information. Sci Data. 2020;7(1):106. http://doi.org/10.1038/s41597-....
3.
Navas-Blanco JR, Dudaryk R. Management of respiratory distress syndrome due to COVID-19 infection. BMC Anesthesiol. 2020;20(1):177. http://doi.org/10.1186/s12871-....
4.
Delikhoon M, Guzman MI, Nabizadeh R, et al. Modes of transmission of severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) and factors influencing on the airborne transmission: a review. Int J Environ Res Public Health. 2021;18(2):395. https://doi.org/10.3390/ ijerph18020395.
5.
Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27:601–615. http://doi.org/10.1038/s41591-....
6.
Kumar VHS, Prasath A, et al. Respiratory failure in an extremely premature neonate with COVID-19. Children (Basel). 2021;8(6):477. http://doi.org/10.3390/childre....
7.
Arango Ferreira C, Correa-Roda M. Acute meningoencephalitis as initial presentation of SARS-CoV-2 infection in pediatrics. Pediatr Infect Dis J. 2020;39(11):e386-e387. https://doi.org/10.1097/ INF.0000000000002885.
8.
Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323(18):1843–1844. http:// doi.org/10.1001/jama.2020.3786.
9.
Chen H, Guo J, Wang C, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395(10226):809–815. https://doi.org/10.1016/S0140-....
10.
Chen W, Lan Y, Yuan X, et al. Detectable 2019-nCoV viral RNA in blood is a strong indicator for the further clinical severity. Emerg Microbes Infect. 2020;9(1):469–473. https://doi.org/10.1080/222217... 1.2020.1732837.
11.
Kumar J, Meena J, Yadav A, et al. SARS-CoV-2 detection in human milk: a systematic review. J Matern Fetal Neonatal Med. 2021:1–8.http://doi.org/10.1080/1476705....
12.
Wagner K, Springer B, Pires VP, et al. Pathogen identification by multiplex lightmix real-time PCR assay in patients with meningitis and culture-negative cerebrospinal fluid specimens. J Clin Microbiol. 2018;56(2):e01492–17. https://doi.org/10.1128/JCM.01....
13.
Rogers T, Sok K, Erickson T, et al. Impact of antibiotic therapy in the microbiological yield of healthcare-associated ventriculitis and meningitis. Open Forum Infect Dis. 2019;6(3):ofz050. http://doi. org/10.1093/ofid/ofz050.
14.
Bellon M, Schweblin C, Lambeng N, et al. Cerebrospinal fluid features in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) reverse transcription polymerase chain reaction (RT-PCR) positive patients. Clin Infect Dis. 2021;73(9):e3102-e3105. https://doi.org/10.1093/cid/ci....\.
15.
Abdel-Mannan O, Eyre M, Löbel U, et al. Neurologic and radiographic findings associated withhttp://doi.org/10.1186/s13052-018-0573-y COVID-19 infection in children. JAMA Neurol. 2020;77(11):1440–1445. https://doi.org/10.1001/jamane....
16.
May M, Daley AJ, Donath S, et al. Australasian study group for neonatal infections. Early onset neonatal meningitis in Australia and New Zealand, 1992–2002. Arch Dis Child Fetal Neonatal Ed. 2005;90: F324-F327. http://doi.org/10.1136/adc.200....
17.
Salmon-Rousseau A, Martins C, Blot M, et al. Comparative review of imipenem/cilastatin versus meropenem. Med Mal Infect. 2020;50(4):316–322. https://doi.org/10.1016/j.medm....
18.
Bryce AN, Doocey R, Handy R. Staphylococcus haemolyticus meningitis and bacteremia in an allogenic stem cell transplant patient. IDCases. 2021;26:e01259. https://doi.org/10.1016/j.idcr....
19.
Chen XK, Shi HY, Leroux S, et al. Penetration of cefotaxime into cerebrospinal fluid in neonates and young infants. Antimicrob Agents Chemother. 2018;62(4):e02448–17. https://doi.org/10.1128/AAC.02...- 17.
20.
O’Leary CK, Jones C, Bryant PA, et al. Feasibility of continuous Infusions of acyclovir. Pediatr Infect Dis J. 2020;39(9):830–832. https:// doi.org/10.1097/INF.0000000000002692.
21.
Schaenman JMD, Ho Dy, Baden LR, et al. Enterovirus infection in immunocompromised hosts. In: Safdar A, editors. Principles and practice of transplant infectious diseases. New York, NY: Springer. https://doi.org/10.1007/978-1-....
22.
Chen F, Zou L, Williams B, et al. Targeting toll-like receptors in sepsis: from bench to clinical trials. Antioxid Redox Signal. 2021;35(15):1324–1339. https://doi.org/10.1089/ars.20....
23.
Ma X, Tian D, Lv W, et al. Anti-inflammatory effects of microRNA-223 on sepsis-induced lung injury in rats by targeting the Toll-like receptor signaling pathway. Exp Ther Med. 2021;22(3):964. http://doi. org/10.3892/etm.2021.10396.
24.
Saravolatz LD, Depcinski S, Sharma M. Molnupiravir and nirmatrelvirritonavir: oral COVID antiviral drugs. Clin Infect Dis. 2022: ciac180. http://doi.org/10.1093/cid/cia....
25.
Painter GR, Natchus MG, Cohen O, et al. Developing a direct acting, orally available antiviral agent in a pandemic: the evolution of molnupiravir as a potential treatment for COVID-19. Curr Opin Virol. 2021;50:17–22. http://doi.org/10.1016/j.covir....
26.
García CAC, Sánchez EBA, Huerta DH, et al. Covid-19 treatmentinduced neuropsychiatric adverse effects. Gen Hosp Psychiatry.2020;67:163–164. http://doi.org/10.1016/j.genho....
27.
Yang K. What do we know about remdesivir drug interactions? Clin Transl Sci. 2020;13(5):842–844. http://doi.org/10.1111/cts.128... AAEM Annals of Agricultural and Environmental Medicine.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.