Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Clinical significance of miR-218-5p and its potential mechanism in acute pulmonary embolism
1
Department of Respiratory Medicine, Dalian University Affiliated Xinhua Hospital, China
2
Department of Cardiovascular Medicine, Dalian University Affiliated Xinhua Hospital, China
Corresponding author
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Acute pulmonary embolism (APE), a life-endangering cardiovascular acute disorder, brings about difficulties in early diagnosis. The aim of the study is to investigate the expression of miR-218-5p, its clinical significance, and mechanism in APE.
Material and methods:
A total of 102 APE patients and 98 healthy controls were recruited, with miR-218-5p levels assayed by qRT-PCR; its diagnostic value was assessed by ROC curves. Pearson correlation and multivariate logistic regression analyzed associations with clinical indicators and independent predictive value. Oxygen-glucose deprivation/reoxygenation (OGD/R)-induced human pulmonary artery endothelial cells (HPAECs) were transfected with miR-218-5p mimic to observe the effects on apoptosis, proliferation, inflammation, and oxidative stress. Bioinformatics and dual-luciferase assays validated miR-218-5p targeting CREB1.
Results:
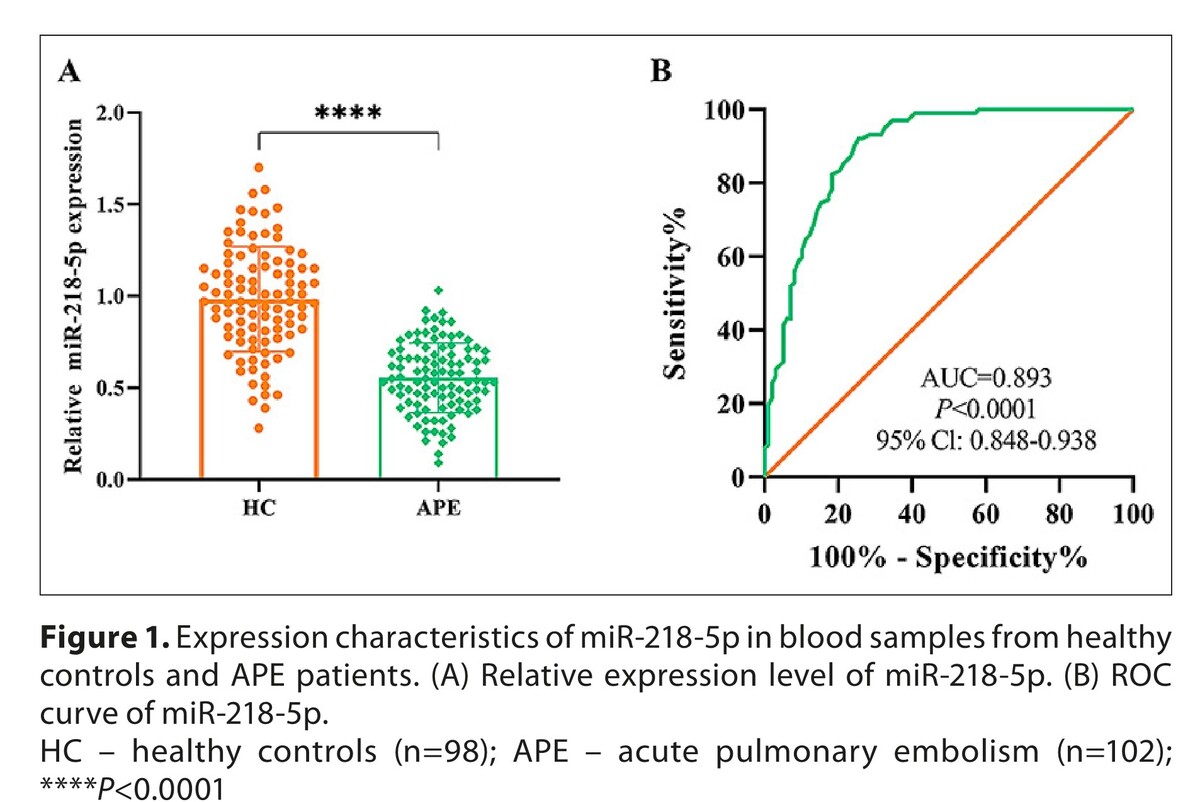
Serum miR-218-5p was significantly downregulated in APE (P<0.0001) with high diagnostic efficacy (AUC=0.893). It positively correlated with D-dimer (r=0.759) and Wells scores (r=0.703; P<0.0001) and was an independent APE risk factor (OR=0.053; P<0.0001). In vitro, miR-218-5p overexpression reduced OGD/R-induced HPAEC apoptosis, promoted proliferation, inhibited inflammation (IL-6, IL-1β, TNF-α) and oxidative stress (MDA, ROS), and restored SOD activity (P<0.01). Mechanistically, miR-218-5p directly targeted CREB1 to suppress its expression.
Conclusions:
miR-218-5p is downregulated in APE, as a potential diagnostic biomarker. It targets CREB1 to modulate apoptosis, inflammation, and oxidative stress, thereby contributing to APE pathogenesis.
Acute pulmonary embolism (APE), a life-endangering cardiovascular acute disorder, brings about difficulties in early diagnosis. The aim of the study is to investigate the expression of miR-218-5p, its clinical significance, and mechanism in APE.
Material and methods:
A total of 102 APE patients and 98 healthy controls were recruited, with miR-218-5p levels assayed by qRT-PCR; its diagnostic value was assessed by ROC curves. Pearson correlation and multivariate logistic regression analyzed associations with clinical indicators and independent predictive value. Oxygen-glucose deprivation/reoxygenation (OGD/R)-induced human pulmonary artery endothelial cells (HPAECs) were transfected with miR-218-5p mimic to observe the effects on apoptosis, proliferation, inflammation, and oxidative stress. Bioinformatics and dual-luciferase assays validated miR-218-5p targeting CREB1.
Results:
Serum miR-218-5p was significantly downregulated in APE (P<0.0001) with high diagnostic efficacy (AUC=0.893). It positively correlated with D-dimer (r=0.759) and Wells scores (r=0.703; P<0.0001) and was an independent APE risk factor (OR=0.053; P<0.0001). In vitro, miR-218-5p overexpression reduced OGD/R-induced HPAEC apoptosis, promoted proliferation, inhibited inflammation (IL-6, IL-1β, TNF-α) and oxidative stress (MDA, ROS), and restored SOD activity (P<0.01). Mechanistically, miR-218-5p directly targeted CREB1 to suppress its expression.
Conclusions:
miR-218-5p is downregulated in APE, as a potential diagnostic biomarker. It targets CREB1 to modulate apoptosis, inflammation, and oxidative stress, thereby contributing to APE pathogenesis.
REFERENCES (36)
1.
Kulka HC, Zeller A, Fornaro J, et al. Acute Pulmonary Embolism–Its Diagnosis and Treatment From a Multidisciplinary Viewpoint. Dtsch Arztebl Int. 2021;118(37):618–28.
2.
Howard L. Acute pulmonary embolism. Clinical medicine (London, England). 2019;19(3):243–7.
3.
Toplis E, Mortimore G. The diagnosis and management of pulmonary embolism. Br J Nurs. 2020;29(1):22–6.
4.
Zhang MW, Shen YJ, Shi J, Yu JG. MiR-223-3p in Cardiovascular Diseases: A Biomarker and Potential Therapeutic Target. Front Cardiovasc Med. 2020;7:610561.
5.
Matveeva NA, Baulina NM, Kiselev IS, et al. [MiRNA miR-375 as a Multifunctional Regulator of the Cardiovascular System]. Mol Biol (Mosk). 2022;56(3):418–27.
6.
Vishnoi A, Rani S. miRNA Biogenesis and Regulation of Diseases: An Updated Overview. Methods Mol Biol. 2023;2595:1–12.
7.
Li Y, Shao J, Song J, et al. MiR-34a-3p suppresses pulmonary vascular proliferation in acute pulmonary embolism rat by targeting DUSP1. Biosci Rep. 2022;42(1).
8.
Chen H, Ma Q, Zhang J, et al. miR-106b-5p modulates acute pulmonary embolism via NOR1 in pulmonary artery smooth muscle cells. Int J Mol Med. 2020;45(5):1525–33.
9.
Liu T, Kang J, Liu F. Plasma Levels of microRNA-221 (miR-221) are Increased in Patients with Acute Pulmonary Embolism. Med Sci Monit. 2018;24:8621–6.
10.
Shan Y, Chen Y, Brkić J, et al. miR-218-5p Induces Interleukin-1β and Endovascular Trophoblast Differentiation by Targeting the Transforming Growth Factor β-SMAD2 Pathway. Frontiers in endocrinology. 2022;13:842587.
11.
Hu S, Li Z, Lutz H, et al. Dermal exosomes containing miR-218-5p promote hair regeneration by regulating β-catenin signaling. Sci Adv. 2020;6(30):eaba1685.
12.
Gu J, Ge X, You A, et al. miR-218-5p inhibits the malignant progression of glioma via targeting TCF12. Tumori. 2022;108(4):338–46.
13.
Felix G, Ferreira E, Ribeiro A, et al. Predictors of cancer in patients with acute pulmonary embolism. Thromb Res. 2023;230:11–7.
14.
Henkin S, Ujueta F, Sato A, Piazza G. Acute Pulmonary Embolism: Evidence, Innovation, and Horizons. Curr Cardiol Rep. 2024;26(11):1249–64.
15.
Ho PTB, Clark IM, Le LTT. MicroRNA-Based Diagnosis and Therapy. Int J Mol Sci. 2022;23(13).
16.
Sobrero M, Montecucco F, Carbone F. Circulating MicroRNAs for Diagnosis of Acute Pulmonary Embolism: Still a Long Way to Go. Biomed Res Int. 2022;2022:4180215.
17.
Liu Y, Xie M, Gao X, Liu R. Predictive Value of Circulating microRNA-134 Levels for Early Diagnosis of Acute Pulmonary Embolism: Meta-analysis. J Cardiovasc Transl Res. 2021;14(4):744–53.
18.
Kessler T, Erdmann J, Vilne B, et al. Serum microRNA-1233 is a specific biomarker for diagnosing acute pulmonary embolism. J Transl Med. 2016;14(1):120.
19.
Fuchs E, Asakly S, Karban A, Tzoran I. Age-Adjusted Cutoff D-Dimer Level to Rule Out Acute Pulmonary Embolism: A Validation Cohort Study. Am J Med. 2016;129(8):872–8.
20.
Jiao X, Zhang Y, Kuang T, et al. The 3-level Wells score combined with D-dimer can accurately diagnose acute pulmonary embolism in hospitalized patients with acute exacerbation of COPD: A multicentre cohort study. Int J Cardiol Heart Vasc. 2024;55:101533.
21.
Zhao Y, Cheng Y, Wang H, et al. The Legend score synthesizes Wells, PERC, Geneva, D-dimer and predicts acute pulmonary embolism prior to imaging tests. Pulmonology. 2025;31(1):2416828.
22.
Shen JH, Chen HL, Chen JR, et al. Comparison of the Wells score with the revised Geneva score for assessing suspected pulmonary embolism: a systematic review and meta-analysis. J Thromb Thrombolysis. 2016;41(3):482–92.
23.
Siddiqui F, Tafur A, Hussain M, et al. The prognostic value of blood cellular indices in pulmonary embolism. Am J Hematol. 2024;99(9):1704–11.
24.
Chen H, Liu D, Ge L, et al. Catestatin prevents endothelial inflammation and promotes thrombus resolution in acute pulmonary embolism in mice. Biosci Rep. 2019;39(11).
25.
Hu M, Qiu H, He T, Zhong M. Effect of miRNA-218-5p on Proliferation, Migration, Apoptosis and Inflammation of Vascular Smooth Muscle Cells in Abdominal Aortic Aneurysm and Extracellular Matrix Protein. Iran J Public Health. 2022;51(11):2494–503.
26.
Wang S, Wang F, Ren J, Yang H. LncRNA NEAT1 Promotes Vascular Endothelial Cell Dysfunction via miR-218-5p/GAB2 and Serves as a Diagnostic Biomarker for Deep Vein Thrombosis. Clin Appl Thromb Hemost. 2023;29:10760296231179447.
27.
Gu X, Sun X, Yu Y, Li L. MiR-218-5p promotes trophoblast infiltration and inhibits endoplasmic reticulum/oxidative stress by reducing UBE3A-mediated degradation of SATB1. J Cell Commun Signal. 2023;17(3):993–1008.
28.
Jian X, Yang D, Wang L, Wang H. CREB1 Silencing Protects Against Inflammatory Response in Rats with Deep Vein Thrombosis Through Reducing RPL9 Expression and Blocking NF-κB Signaling. J Cardiovasc Transl Res. 2024;17(3):570–84.
29.
Kim MJ, Choi HJ, Kang HY, et al. CREB1 controls mitochondrial dysfunction in 1-nitropyrene-mediated apoptosis of human bronchial cells and lung injury. Sci Total Environ. 2025;968:178877.
30.
Wu Y, Sun X, Cui G, Wang S. Mir-150-5p distinguishes acute pulmonary embolism, predicts the occurrence of pulmonary arterial hypertension, and regulates ox-LDL-induced endothelial cell injury. Hereditas. 2024;161(1):33.
31.
Shen S, Yan B, Li Y, Jiang J. Berberine alleviates acute embolism-induced lung inflammation and injury by suppressing miR-22-5p expression and modulating the TLR4/NF-κB signaling pathway. Toxicol Appl Pharmacol. 2025;505:117563.
32.
Deng R, Chen J, Qin J, et al. Effect of circFOXM1/miR-218-5p on the proliferation, apoptosis and migration of glioma cells. Cell Mol Biol (Noisy-le-grand). 2024;70(4):191–5.
33.
Zhao Z, Cheng J, Sun W, et al. The LINC01176-miR-218-5p-IL-36G Network is Responsible for the Pathogenesis of Psoriasis by Promoting Inflammation. Clin Cosmet Investig Dermatol. 2024;17:1–12.
34.
Glazier CR, Baciewicz FA, Jr. Epidemiology, Etiology, and Pathophysiology of Pulmonary Embolism. Int J Angiol. 2024;33(2):76–81.
35.
Zhu L, Dong H, Li L, Liu X. The Mechanisms of Sepsis Induced Coagulation Dysfunction and Its Treatment. J Inflamm Res. 2025;18:1479–95.
36.
Hu Z, Zhou G. CREB1 Transcriptionally Activates LTBR to Promote the NF-κB Pathway and Apoptosis in Lung Epithelial Cells. Comput Math Methods Med. 2022;2022:9588740.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.