Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Analysis of the health-promoting properties of herbal mixture B9 with the addition of whey powder
1
Department of Dietetics and Nutrition Education, Medical University, Lublin, Poland
2
Pharmaceutical Biotechnology Department, Medical University, Łódż, Poland
3
Institute of Food Technology and Analysis, Lodz University of Technology, Łódż, Poland
4
Department of Biology and Pharmaceutical Botany, Medical University, Łódź, Poland
5
Department of Pharmaceutical Microbiology and Microbiological Diagnostic, Medical University, Poland
6
Mon-Intra Co., Ltd, Mongolia
7
Department of Diagnostic Immunology, Pomeranian Medical University, Szczecin, Poland
8
Department of Experimental Immunology, Medical University, Lublin, Poland
9
School of Pharmacy, National University of Medical Sciences, Mongolia
Corresponding author
Ewa Dudzińska
Department of Dietetics and Nutrition Education,, Medical University of Lublin, Chodzki 7 str., 20-059 Lublin, Poland, Poland
Department of Dietetics and Nutrition Education,, Medical University of Lublin, Chodzki 7 str., 20-059 Lublin, Poland, Poland
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Irritable bowel syndrome (IBS) is a chronic functional gastrointestinal disorder characterized by recurrent disturbances in bowel habits and abdominal discomfort. Modulation of the gut microbiota and oxidative balance using plant-derived metabolites represents a promising supportive strategy for functional gastrointestinal disorders. The aim of this study is to evaluate the in vitro biological activity and cytocompatibility of the herbal mixture B9 supplemented with whey protein in the context of intestinal health.
Material and methods:
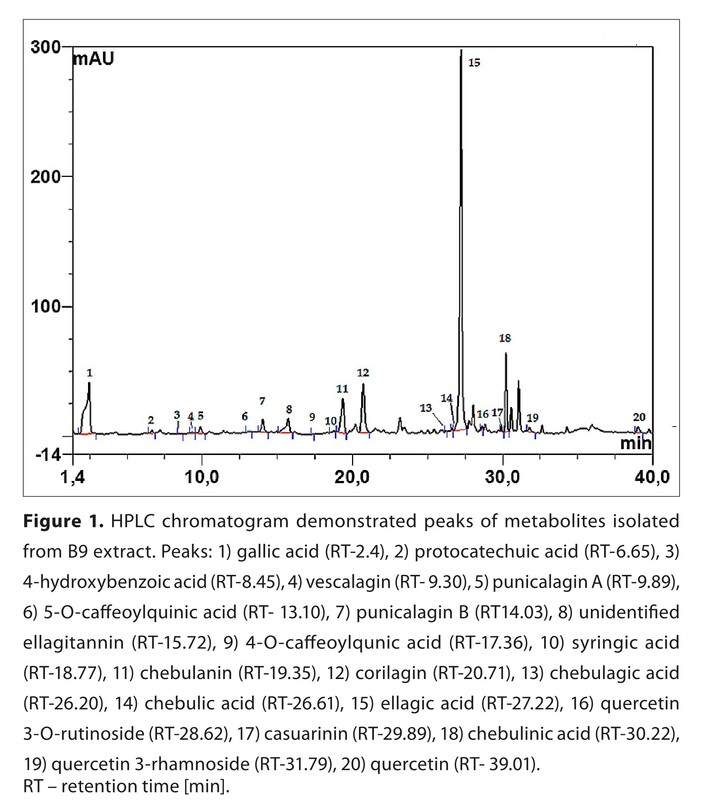
The phenolic profile of the B9 extract was characterized using UHPLC-ESI-MS/MS and included ellagic acid, gallic acid, chebulanin, chebulic acid, corilagin, and quercetin. Antioxidant activity was assessed using the DPPH assay; the B9 preparation exhibited strong radical scavenging activity (EC50 = 4.13 µg/mL), nearly six times higher than butylated hydroxytoluene (BHT) and comparable to Trolox. Antioxidant effects were further evaluated by measuring protein carbonyl group levels in Enterococcus faecalis ATCC 29212. Cytocompatibility was assessed by measuring the metabolic activity of normal intestinal epithelial cells (CCD 841 CoTr) using the MTT assay.
Results:
The B9 preparation significantly reduced protein carbonyl levels in E. faecalis, indicating antioxidative activity. Co-treatment with whey increased metabolic activity of intestinal epithelial cells, with the strongest effects observed at 3.0% and 5.0% (v/v) of the B9 extract combined with 7.5% (v/v) of whey, indicating good cytocompatibility.
Conclusions:
In conclusion, the B9 herbal preparation enriched with whey exhibits pronounced antioxidant activity and favourable cytocompatibility toward normal intestinal epithelial cells in vitro, supporting its further investigation in the context of intestinal health and functional gastrointestinal disorders such as IBS.
Irritable bowel syndrome (IBS) is a chronic functional gastrointestinal disorder characterized by recurrent disturbances in bowel habits and abdominal discomfort. Modulation of the gut microbiota and oxidative balance using plant-derived metabolites represents a promising supportive strategy for functional gastrointestinal disorders. The aim of this study is to evaluate the in vitro biological activity and cytocompatibility of the herbal mixture B9 supplemented with whey protein in the context of intestinal health.
Material and methods:
The phenolic profile of the B9 extract was characterized using UHPLC-ESI-MS/MS and included ellagic acid, gallic acid, chebulanin, chebulic acid, corilagin, and quercetin. Antioxidant activity was assessed using the DPPH assay; the B9 preparation exhibited strong radical scavenging activity (EC50 = 4.13 µg/mL), nearly six times higher than butylated hydroxytoluene (BHT) and comparable to Trolox. Antioxidant effects were further evaluated by measuring protein carbonyl group levels in Enterococcus faecalis ATCC 29212. Cytocompatibility was assessed by measuring the metabolic activity of normal intestinal epithelial cells (CCD 841 CoTr) using the MTT assay.
Results:
The B9 preparation significantly reduced protein carbonyl levels in E. faecalis, indicating antioxidative activity. Co-treatment with whey increased metabolic activity of intestinal epithelial cells, with the strongest effects observed at 3.0% and 5.0% (v/v) of the B9 extract combined with 7.5% (v/v) of whey, indicating good cytocompatibility.
Conclusions:
In conclusion, the B9 herbal preparation enriched with whey exhibits pronounced antioxidant activity and favourable cytocompatibility toward normal intestinal epithelial cells in vitro, supporting its further investigation in the context of intestinal health and functional gastrointestinal disorders such as IBS.
FUNDING
This work was supported by the Medical University of Lublin,
Grant number DS 675.
REFERENCES (30)
1.
Dudzińska E, Grabrucker AM, Kwiatkowski P, et al. The Importance of Visceral Hypersensitivity in Irritable Bowel Syndrome-Plant Metabolites in IBS Treatment. Pharmaceuticals (Basel). 2023 Oct 3;16(10):1405. doi: 10.3390/ph16101405. PMID: 37895876; PMCID: PMC10609912.
2.
Di Rosa C, Altomare A, Terrigno V, et al. Constipation-Predominant Irritable Bowel Syndrome (IBS-C): Effects of Different Nutritional Patterns on Intestinal Dysbiosis and Symptoms. Nutrients. 2023 Mar 28;15(7):1647. doi:10.3390/nu15071647. PMID: 37049488; PMCID: PMC10096616.
3.
Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30:492–506. https://doi.org/10.1038.
4.
Chen Y, Feng S, Li Y, et al. Zhang S. Gut microbiota and intestinal immunity-A crosstalk in irritable bowel syndrome. Immunology. 2024 May;172(1):1–20. doi:10.1111/imm.13749. Epub 2024 Jan 4. PMID: 38174581.
5.
Jayasinghe M, Karunanayake V, Mohtashim A, et al. The Role of Diet in the Management of Irritable Bowel Syndrome: A Comprehensive Review. Cureus. 2024 Feb 15;16(2):e54244. doi:10.7759/cureus.54244. PMID: 38496157; PMCID: PMC10944297.
6.
Mazzawi T. Gut Microbiota Manipulation in Irritable Bowel Syndrome. Microorganisms. 2022 Jun 30;10(7):1332. doi:10.3390/microorganisms10071332. PMID: 35889051; PMCID: PMC9319495.
7.
Nguyen DN, Sangild PT, Li Y, et al. Processing of whey modulates proliferative and immune functions in intestinal epithelial cells. J Dairy Sci. 2016 Feb;99(2):959–969. doi:10.3168/jds.2015-9965. Epub 2015 Dec 17. PMID: 26709184.
8.
Batjargal A, Solek P, Kukula-Koch W, et al. Gurgem-7 toxicity assessment: Regulation of cell survival or death by traditional Mongolian prescription. Ecotoxicol Environ Saf. 2022 Jul 1;239:113660. doi: 10.1016/j.ecoenv.2022.113660. Epub 2022 May 20. PMID: 35605329.
9.
Oracz J, Żyżelewicz D, Pacholczyk-Sienicka B. UHPLC-DAD-ESI-HRMS/MS profile of phenolic compounds in northern red oak (Quercus rubra L., syn. Q. borealis F. Michx) seeds and its transformation during thermal processing. Industrial Crops & Products. 2022;189:115860. https://doi.org/10.1016/j.indc....
10.
Re R, Pellegrini N, Proteggente A, et al. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999 May;26(9–10):1231–7. doi:10.1016/s0891-5849(98)00315-3. PMID: 10381194.
11.
Brand–Williams W, Cuvelier ME, Berset C. Use of a free radical methods evaluate antioxidant activity. Lebensm Wiss Technol. 1995;28:25–30.
12.
Pulido R, Bravo L, Saura-Calixto F. Antioxidant activities of dietary phenols are determined by a modified ferric reducing/antioxidant power assay. J Agric Food Chem. 2000;46:3396–3402.
13.
Georgiou CD, Zisimopoulos D, Argyropoulou V, et al. Protein and cell wall polysaccharide carbonyl determination by a neutral pH 2,4-dinitrophenylhydrazine-based photometric assay. Redox Biol. 2018 Jul;17:128–142. doi:10.1016/j.redox.2018.04.010. Epub 2018 Apr 10. PMID: 29684819; PMCID: PMC6006683.
14.
Estévez M, Díaz-Velasco S, Martínez R. Protein carbonylation in food and nutrition: a concise update. Amino Acids. 2022 Apr;54(4):559–573. doi:10.1007/s00726-021-03085-6. Epub 2021 Oct 20. PMID: 34669011; PMCID: PMC9117389.
15.
Munteanu G, Apetrei C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int J Mol Sci. 2021;22:3380.
16.
Dhingra AK, Chopra B, Grewal AS, et al. Pharmacological properties of Chebulinic acid and related ellagitannins from nature: An emerging contemporary bioactive entity. Pharmacol Res – Modern Chinese Med. 2022;5:100163 https://doi.org/10.1016/j.prmc....
17.
Bag A, Bhattacharyya SK, Chattopadhyay RR. The development of Terminalia chebula Retz. (Combretaceae) in clinical research. Asian Pac J Trop Biomed. 2013;3(3):244–252.
18.
Li X, Deng Y, Zheng Z, et al. Corilagin, a promising medicinal herbal agent. Biomed Pharmacother. 2018 Mar;99:43–50. doi:10.1016/j.biopha.2018.01.030. Epub 2018 Jan 8. PMID: 29324311.
19.
Bai J, Zhang Y, Tang C, et al. Gallic acid: Pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomed Pharmacother. 2021;133:110985.
20.
Zamudio-Cuevas Y, Andonegui-Elguera MA, Aparicio-Juárez A, et al. The enzymatic poly(gallic acid) reduces pro-inflammatory cytokines in vitro, a potential application in inflammatory diseases. Inflammation. 2021;44(1):174–185.
21.
Dudzińska E, Gryzinska M, Kocki J. Single Nucleotide Polymorphisms in Selected Genes in Inflammatory Bowel Disease. Biomed Res Int. 2018 Dec 17;2018:6914346. doi:10.1155/2018/6914346. PMID: 30648106; PMCID: PMC6311883.
22.
Somade OT, Oyinloye BE, Ajiboye BO, et al. Syringic acid demonstrates an anti-inflammatory effect via modulation of the NF-κB-iNOS-COX-2 and JAK-STAT signaling pathways in methyl cellosolve-induced hepato-testicular inflammation in rats. Biochem Biophys Rep. 2023 May 5;34:101484. doi:10.1016/j.bbrep.2023.101484. PMID: 37197735; PMCID: PMC10184048.
23.
Bartel I, Mandryk I, Horbańczuk JO, et al. Nutraceutical Properties of Syringic Acid in Civilization Diseases-Review. Nutrients. 2023 Dec 19;16(1):10.
24.
Venusova E, Kolesarova A, Horky P, et al. Physiological and Immune Functions of Punicalagin. Nutrients. 2021 Jun 23;13(7):2150. doi:10.3390/nu13072150. PMID: 34201484; PMCID: PMC8308219.
25.
Wang G, Wang Y, Yao L, et al. Pharmacological Activity of Quercetin: An Updated Review. Evid Based Complement Alternat Med. 2022 Dec 1;2022:3997190. doi:10.1155/2022/3997190. PMID: 36506811; PMCID: PMC9731755.
26.
Kao PHN, Kline KA. Dr. Jekyll and Mr. Hide: How Enterococcus faecalis Subverts the Host Immune Response to Cause Infection. J Mol Biol. 2019 Jul 26;431(16):2932–2945. doi:10.1016/j.jmb.2019.05.030. Epub 2019 May 25. PMID: 31132360.
27.
Huycke MM, Abrams V, Moore DR. Enterococcus faecalis produces extracellular superoxide and hydrogen peroxide that damages colonic epithelial cell DNA. Carcinogenesis. 2002;23(3):529–36. doi:10.1093/carcin/23.3.529. PMID: 11895869.
28.
Million M, Tidjani Alou M, Khelaifia S, et al. Increased Gut Redox and Depletion of Anaerobic and Methanogenic Prokaryotes in Severe Acute Malnutrition. Sci Rep. 2016;6:26051. doi:10.1038/srep26051. Retraction in: Sci Rep. 2023;13(1):18589. PMID: 27183876; PMCID: PMC4869025.
29.
Dallas DC, Guerrero A, Parker EA, et al. Peptidomic profile of milk of Holstein cows at peak lactation. J Agric Food Chem. 2014 Jan 8;62(1):58–65. doi:10.1021/jf4040964. Epub 2013 Dec 23.
30.
Miallot R, Millet V, Galland F, et al. The vitamin B5/coenzyme A axis: A target for immunomodulation? Eur J Immunol. 2023;53(10):e2350435.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.