Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Toxocara infection in children with type 1 diabetes mellitus in Iran: a case-control seroprevalence study
1
Department of Pediatrics, School of Medicine, Imam Ali Hospital, Alborz University of Medical Sciences, Karaj, Iran
2
Department of Parasitology and Mycology, School of Medicine, Alborz University of Medical Sciences, Karaj, Iran
3
Center for Research of Endemic Parasites of Iran, Tehran University of Medical Sciences, Tehran, Iran
4
Department of Microbiology, School of Medicine, Alborz University of Medical Sciences, Karaj, Iran
5
Social Determinants of Health Research Center, Alborz University of Medical Sciences, Karaj, Iran
Corresponding author
Mohammad Zibaei
Department of Parasitology, Alborz University of Medical Sciences, Square Talaghani, North Talaghani Blvd, 3149779453, Karaj, Iran
Department of Parasitology, Alborz University of Medical Sciences, Square Talaghani, North Talaghani Blvd, 3149779453, Karaj, Iran
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Parasitic diseases and subsequent immunity against helminth infections involves profound changes in both the innate and adaptive immune compartments, which can have a protective effect in inflammation and autoimmunity diseases such as type 1 diabetes mellitus. The aim of the study is to evaluate the prevalence of anti-Toxocara antibodies and associated risk factors among diabetic children in a case-control study.
Material and methods:
A total of 105 blood samples from diabetic children, and 101 non-diabetic controls attending the Comprehensive Imam Ali Children’s Hospital, Alborz University of Medical Sciences in Karaj, Iran, were collected and tested by ELISA for anti-Toxocara antibodies. A questionnaire administered by direct interviews was used to collect socio-demographic information and data on associated risk factors.
Results:
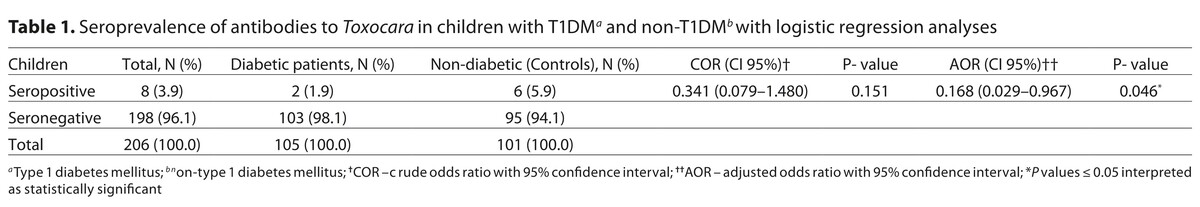
The total seroprevalence of anti-Toxocara antibodies was significantly higher in healthy control individuals (5.9%) than in diabetic children (1.9%) (COR: 0.341, 95% CI: 0.079–1.480; AOR: 0.168, 95% CI: 0.029–0.967). Furthermore, seropositivity for toxocariasis was higher in the uncontrolled diabetic group than in controlled diabetic children, this was statistically significant (P = 0.046). Female participants (62.5%) were more susceptible to Toxocara infection. Also, living in rural areas (62.5%), illiteracy (75.0%), and contact with non-domesticated animals and/or soil (75.5%) were found to be the highest risk factors for toxocariasis in the studied children.
Conclusions:
The finding of the study supports the hypothesis of an association between parasitic helminths infections and diabetes mellitus, and that T1DM can be considered as a stimulus in regulating and modulating the immune system in toxocariasis. More accurate results could be obtained from a larger than necessary sample better representative of the population.
Parasitic diseases and subsequent immunity against helminth infections involves profound changes in both the innate and adaptive immune compartments, which can have a protective effect in inflammation and autoimmunity diseases such as type 1 diabetes mellitus. The aim of the study is to evaluate the prevalence of anti-Toxocara antibodies and associated risk factors among diabetic children in a case-control study.
Material and methods:
A total of 105 blood samples from diabetic children, and 101 non-diabetic controls attending the Comprehensive Imam Ali Children’s Hospital, Alborz University of Medical Sciences in Karaj, Iran, were collected and tested by ELISA for anti-Toxocara antibodies. A questionnaire administered by direct interviews was used to collect socio-demographic information and data on associated risk factors.
Results:
The total seroprevalence of anti-Toxocara antibodies was significantly higher in healthy control individuals (5.9%) than in diabetic children (1.9%) (COR: 0.341, 95% CI: 0.079–1.480; AOR: 0.168, 95% CI: 0.029–0.967). Furthermore, seropositivity for toxocariasis was higher in the uncontrolled diabetic group than in controlled diabetic children, this was statistically significant (P = 0.046). Female participants (62.5%) were more susceptible to Toxocara infection. Also, living in rural areas (62.5%), illiteracy (75.0%), and contact with non-domesticated animals and/or soil (75.5%) were found to be the highest risk factors for toxocariasis in the studied children.
Conclusions:
The finding of the study supports the hypothesis of an association between parasitic helminths infections and diabetes mellitus, and that T1DM can be considered as a stimulus in regulating and modulating the immune system in toxocariasis. More accurate results could be obtained from a larger than necessary sample better representative of the population.
REFERENCES (25)
1.
Olanipekun JA, Babatunde JO. Towards reducing the burden of global environmental related health problems in the 21st century. J Educ Prac. 2016;7(32):57–64.
2.
Zibaei M, Sadjjadi SM, Uga S. Experimental Toxocara cati infection in gerbils and rats. Korean J Parasitol. 2010;48(4):331–333. https://doi.org/10.3347/kjp.20....
3.
Despommier D. Toxocariasis: clinical aspects, epidemiology, medical ecology, and molecular aspects. Clin Microbiol Rev. 2003;16(2):265–272. https://doi.org/10.1128/CMR.16....
4.
Delai RR, Freitas AR, Kmetiuk LB, et al. One Health approach on human seroprevalence of anti-Toxocara antibodies, Toxocara spp. eggs in dogs and sand samples between seashore mainland and island areas of southern Brazil. One Health. 2021;13:100353. https://doi.org/10.1016/j.oneh....
5.
Eslahi AV, Badri M, Khorshidi A, et al. Prevalence of Toxocara and Toxascaris infection among human and animals in Iran with meta-analysis approach. BMC Infect Dis. 2020;20(1):20. https://doi.org/10.1186/s12879....
6.
Chen J, Liu Q, Liu GH, et al. Toxocariasis: a silent threat with a progressive public health impact. Infect Dis Poverty. 2018;7(1):59. https://doi.org/10.1186/s40249....
7.
Jakobsen OAJ, Szereday L. The “Three Amigos” lurking behind type 1 diabetes: Hygiene, gut microbiota and viruses. Acta Microbiol Immunol Hung. 2018;65(4):421–438. https://doi.org/10.1556/030.65....
8.
Zaccone P, Hall SW. Helminth infection and type 1 diabetes. Rev Diabet Stud. 2012;9(4):272–286. https://doi.org/10.1900/RDS.20....
9.
Liu Q, Sundar K, Mishra PK, et al. Helminth infection can reduce insulitis and type 1 diabetes through CD25- and IL-10-independent mechanisms. Infect Immun. 2009;77(12):5347–558. https://doi.org/10.1128/IAI.01....
10.
Zaccone P, Fehérvári Z, Jones FM, et al. Schistosoma mansoni antigens modulate the activity of the innate immune response and prevent onset of type 1 diabetes. Eur J Immunol. 2003;33(5):1439–1449. https://doi.org/10.1002/eji.20....
11.
Bruschi F, Ashour DS, Othman AA. Trichinella-induced immunomodulation: Another tale of helminth success. Food Waterborne Parasitol. 2022;27:e00164. https://doi.org/10.1016/j.fawp....
12.
Park HY, Lee SU, Huh S, Kong Y, Magnaval JF. A seroepidemiological survey for toxocariasis in apparently healthy residents in Gangwon-do, Korea. Korean J Parasitol. 2002;40(3):113–117. https://doi.org/10.3347/kjp.20....
13.
Raissi V, Sohrabi Z, Getso M, et al. Risk factors and prevalence of toxocariasis in pregnant women and diabetic patients compared to healthy adults in Ilam province, western Iran. EXCLI J. 2018;17:983–988. https://doi.org/10.17179/excli....
14.
Zibaei M, Bahadory S, Saadati H, Pourrostami K, Firoozeh F, Foroutan M. Intestinal parasites and diabetes: A systematic review and meta-analysis. New Microbes New Infect. 2022;51:101065. https://doi.org/10.1016/j.nmni....
15.
Zubarev VS, Kardakov IuI. Opisthorchiasis, the pancreas and diabetes mellitus. Med Parazitol (Mosk). 1990;(2):55–57.
16.
Zaccone P, Burton OT, Gibbs S, et al. Immune modulation by Schistosoma mansoni antigens in NOD mice: effects on both innate and adaptive immune systems. J Biomed Biotechnol. 2010; 2010:795210. https://doi.org/10.1900/RDS.20....
17.
Maizels RM, McSorley HJ. Regulation of the host immune system by helminth parasites. J Allergy Clin Immunol. 2016;138(3):666–675. https://doi.org/10.1016/j.jaci....
18.
Saunders KA, Raine T, Cooke A, Lawrence CE. Inhibition of autoimmune type 1 diabetes by gastrointestinal helminth infection. Infect Immun. 2007;75(1):397–407. https://doi.org/10.1128/IAI.00....
19.
Elnadi NA, Hassanien HA, Ahmad AM, Abd Ellah AK. Intestinal parasites in diabetic patients in Sohag University Hospitals, Egypt. J Egypt Soc Parasitol. 2015;45(2):443–449. https://doi.org/10.12816/00175....
20.
Waly WR, Ismail MAGM, Abu-Sarea EY, Abd El Wahab WM. Intestinal parasitic infections and associated risk factors in diabetic patients: A case-control study. J Parasit Dis. 2021;45(4):1106–1113. https://doi.org/10.1007/s12639....
21.
Akinbo FO, Olujobi SO, Omoregie R, Egbe C. Intestinal parasitic infections among diabetes mellitus patients. Biomarkers Genom Med. 2013;5(1–2):44–47. https://doi.org/10.1016/j.gmbh....
22.
Mohtashamipour M, Ghaffari Hoseini SH, Pestehchian N, et al. Intestinal parasitic infections in patients with diabetes mellitus: A case-control study. J Anal Res Clin Med. 2015;3(3):157–163. https://doi.org/10.15171/jarcm....
23.
Zibaei M, Sadjjadi SM, Karamian M, Uga S, Oryan A, Jahadi-Hosseini SH. A comparative histopathology, serology and molecular study, on experimental ocular toxocariasis by Toxocara cati in Mongolian gerbils and Wistar rats. Biomed Res Int. 2013;2013:109580. https://doi.org/10.1155/2013/1....
24.
Zibaei M, Alemi M, Cardillo NM, et al. Human toxocariasis seroprevalence among patients with uveitis in Alborz Province, Iran. Ann Agric Environ Med. 2019;26(1):154–158. https://doi.org/10.26444/aaem/....
25.
Elkholy AA, Omar RE, Elbadawy AM, et al. Investigating the potential link between seroprevalence of Toxoplasma IgG and both types of diabetes mellitus in Benha city, Egypt. Parasitol United J. 2022;15(2):195–201. https://doi.org/10.21608/PUJ.2....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.