Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Chloroquine and hydroxychloroquine – safety profile of potential COVID-19 drugs from the
rheumatologist’s perspective
1
Department of Rheumatology and Internal Medicine, Poznan University of Medical Sciences, Poznań, Poland
2
Department of Clinical Auxology and Pediatric Nursing, Poznan University of Medical Sciences, Poznań, Poland
3
Department of Mathematical Economics, Institute of Informatics and Quantitative Economics, University of Economics
and Business, Poznań, Poland
Corresponding author
Dominik Majewski
Department of Rheumatology and Internal Medicine, Poznań University of Medical Sciences
Department of Rheumatology and Internal Medicine, Poznań University of Medical Sciences
Ann Agric Environ Med. 2021;28(1):122-126
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
The COVID-19 pandemic causes vital concerns due to the lack of proved, effective, and safe therapy. Chloroquine and hydroxychloroquine seem to be useful, but recently serious concerns regarding their adverse events have risen. The aim of the study was to broaden the general perspective of chloroquine and hydroxychloroquine use in COVID-19 treatment, based on an analysis of their current safety profile among patients with rheumatic diseases.
Material and methods:
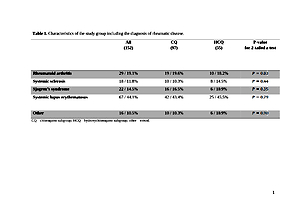
The study was based on a group of 152 patients with rheumatic diseases, aged 20–78 years, treated either with chloroquine or hydroxychloroquine. Analyzed data included age, gender, comorbidities, type of drug, dosage, treatment duration, and reported adverse events. Cases of drug withdrawal related to adverse events were also recorded.
Results:
The dosage was consistent in both groups: 250 mg of chloroquine or 200 mg of hydroxychloroquine daily. 77.6% of patients did not experience any adverse reactions to the treatment. Hydroxychloroquine showed better safety profile, with 10.9% of patients reporting side-ffects, compared to 28.9% in patients treated with chloroquine. The overall incidence of ophthalmic complications was 6.6%. For both drugs, no statistically significant correlation between adverse events and age, chronic heart or liver disease, or hypertension was found.
Conclusions:
Chloroquine and hydroxychloroquine at lower doses, as used in rheumatic diseases, prove to be relatively safe. Data from the literature show that high dosage as recommended in COVID-19 treatment may pose a risk of toxicity and require precise management, but prophylactic, long-term use of lower, safe doses might be a promising solution.
The COVID-19 pandemic causes vital concerns due to the lack of proved, effective, and safe therapy. Chloroquine and hydroxychloroquine seem to be useful, but recently serious concerns regarding their adverse events have risen. The aim of the study was to broaden the general perspective of chloroquine and hydroxychloroquine use in COVID-19 treatment, based on an analysis of their current safety profile among patients with rheumatic diseases.
Material and methods:
The study was based on a group of 152 patients with rheumatic diseases, aged 20–78 years, treated either with chloroquine or hydroxychloroquine. Analyzed data included age, gender, comorbidities, type of drug, dosage, treatment duration, and reported adverse events. Cases of drug withdrawal related to adverse events were also recorded.
Results:
The dosage was consistent in both groups: 250 mg of chloroquine or 200 mg of hydroxychloroquine daily. 77.6% of patients did not experience any adverse reactions to the treatment. Hydroxychloroquine showed better safety profile, with 10.9% of patients reporting side-ffects, compared to 28.9% in patients treated with chloroquine. The overall incidence of ophthalmic complications was 6.6%. For both drugs, no statistically significant correlation between adverse events and age, chronic heart or liver disease, or hypertension was found.
Conclusions:
Chloroquine and hydroxychloroquine at lower doses, as used in rheumatic diseases, prove to be relatively safe. Data from the literature show that high dosage as recommended in COVID-19 treatment may pose a risk of toxicity and require precise management, but prophylactic, long-term use of lower, safe doses might be a promising solution.
REFERENCES (27)
1.
Liu J, Cao R, Xu M, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020; 6: 6–9. http://doi.org/10.1038/s41421-....
2.
Savarino A, Boelaert JR, Cassone A, et al. Effects of chloroquine on viral infections: An old drug against today’s diseases? Lancet Infect Dis. 2003; 3: 722–727. http://doi.org/10.1016/S1473-3....
3.
Principi N, Esposito S. Chloroquine or hydroxychloroquine for prophylaxis of COVID-19. Lancet Infect Dis. 2020. [published online ahead of print] https://doi.org/10.1016/s1473-....
4.
Savarino A. Use of chloroquine in viral diseases. Lancet Infect Dis. 2011; 11: 653–654. https://doi.org/10.1016/S1473-....
5.
Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care 2020; 57: 279–283. https://doi.org/10.1016/j.jcrc....
6.
Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020; 30: 269–271. https://doi.org/10.1038/s41422....
7.
Zhou D, Dai SM, Tong Q. COVID-19: a recommendation to examine the effect of hydroxychloroquine in preventing infection and progression. J Antimicrob Chemother. 2020; 75: 1667–1670. https://doi.org/10.1093/jac/dk....
8.
Yao X, Ye F, Zhang M, et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020. [published online ahead of print] https://doi.org/10.1093/cid/ci....
9.
Gendrot M, Javelle E, Le Dault E, et al. Chloroquine as a prophylactic agent against COVID-19? Int J Antimicrob Agents 2020; 55: 105980. https://doi.org/10.1016/j.ijan....
10.
Mackenzie AH. Dose refinements in long-term therapy of rheumatoid arthritis with antimalarials. Am J Med. 1983; 75: 40–45. https://doi.org/10.1016/0002-9....
11.
Schrezenmeier E, Dörner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol. 2020; 16: 155–166. https://doi.org/10.1038/s41584....
12.
Ruiz-Irastorza G, Olivares N, Ruiz-Arruza I, et al. Predictors of major infections in systemic lupus erythematosus. Arthritis Res Ther. 2009; 11: R109. https://doi.org/10.1186/ar2764.
13.
Rempenault C, Combe B, Barnetche T, et al. Metabolic and cardiovascular benefits of hydroxychloroquine in patients with rheumatoid arthritis: A systematic review and meta-Analysis. Ann Rheum Dis. 2018; 77: 98–103. https://doi.org/10.1136/annrhe....
14.
South AM, Diz DI, Chappell MC. COVID-19, ACE2, and the cardiovascular consequences. Am J Physiol Heart Circ Physiol. 2020; 318: H1084-H1090. https://doi.org/10.1152/ajphea....
15.
Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020; 8: 475–481. https://doi.org/10.1016/S2213-....
16.
Prajapati S, Sharma M, Kumar A, et al. An update on novel COVID-19 pandemic: a battle between humans and virus. Eur Rev Med Pharmacol Sci. 2020; 24: 5819–5829. https://doi.org/10.26355/eurre....
17.
Gao J, Tian Z, Yang X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020; 14: 72–73. https://doi.org/10.5582/BST.20....
18.
Gao J, Hu S. Update on use of chloroquine/hydroxychloroquine to treat coronavirus disease 2019 (COVID-19). Biosci Trends. 2020; 14: 156–158. https://doi.org/10.5582/bst.20....
19.
Wong YK, Yang J, He Y. Caution and clarity required in the use of chloroquine for COVID-19. Lancet Rheumatol. 2020; 2: e255. https://doi.org/10.1016/S2665-....
20.
Mercuro NJ, Yen CF, Shim DJ, et al. Risk of QT Interval Prolongation Associated With Use of Hydroxychloroquine With or Without Concomitant Azithromycin Among Hospitalized Patients Testing Positive for Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020; e201834. https://doi.org/10.1001/jamaca....
21.
Bessiere F, Roccia H, Deliniere A, et al. Assessment of QT Intervals in a Case Series of Patients With Coronavirus Disease 2019 (COVID-19) Infection Treated With Hydroxychloroquine Alone or in Combination With Azithromycin in an Intensive Care Unit. JAMA Cardiol. 2020; e201787. https://doi.org/10.1001/jamaca....
22.
Flisiak R, Horban A, Jaroszewicz J, et al. Recommendations of management in SARS-CoV-2 infection of the Polish Association of Epidemiologists and Infectiologists. Pol Arch Intern Med. 2020; 130: 352–357. https://doi.org/10.20452/pamw.....
23.
Ektorp E. Death threats after a trial on chloroquine for COVID-19. Lancet Infect Dis. 2020; 20: 661. https://doi.org/10.1016/S1473-....
24.
Borba MGS, Val FFA, Sampaio VS, et al. Effect of High vs Low Doses of Chloroquine Diphosphate as Adjunctive Therapy for Patients Hospitalized With Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection: A Randomized Clinical Trial. JAMA Netw Open 2020; 3: e208857. https://doi.org/10.1001/jamane....
25.
Spinelli FR, Ceccarelli F, Di Franco M, et al. To consider or not antimalarials as a prophylactic intervention in the SARS-CoV-2 (Covid-19) pandemic. Ann Rheum Dis. 2020; 79: 666–667. https://doi.org/10.1136/annrhe....
26.
Spinelli FR, Moscarelli E, Ceccarelli F, et al. Treating lupus patients with antimalarials: analysis of safety profile in a single-center cohort. Lupus 2018; 27: 1616–1623. https://doi.org/10.1177/096120....
27.
Sanders JM, Monogue ML, Jodlowski TZ, et al. Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020. [published online ahead of print] https://doi.org/10.1001/jama.2....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.