Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Prediction value of soluble urokinase plasminogen activator receptor (suPAR) in COVID-19 patients – a systematic review and meta-analysis
1
Central Clinical Hospital of the Ministry of Interior and Administration, Warsaw, Poland
2
Polish Society of Disaster Medicine, Poland
3
Medical University, Bialystok, Poland
4
Baylor College of Medicine, Houston, United States
5
Medical University, Warsaw, Poland
6
The Institute of Environmental Protection – National Research Institute (IEP-NRI), Warsaw, Poland
7
Institute of Rural Health, Lublin, Poland
8
Maria Sklodowska-Curie Medical University, Warsaw, Poland
Ann Agric Environ Med. 2023;30(1):142-147
KEYWORDS
TOPICS
- Biological agents posing occupational risk in agriculture, forestry, food industry and wood industry and diseases caused by these agents (zoonoses, allergic and immunotoxic diseases)
- State of the health of rural communities depending on various factors: social factors, accessibility of medical care, etc.
ABSTRACT
Introduction and objective:
In COVID-19, the rapid prediction of the severity of a patient’s condition using modern biomarkers can accelerate the implementation of appropriate therapy, and thus improve the patient’s prognosis.
Material and methods:
A meta-analysis was conducted of data available in the literature on the differences in baseline suPAR blood concentration between patients (1) who tested positive and negative for COVID-19, (2) who had severe and non-severe COVID-19, and (3) COVID-19 survivors and non-survivors.
Results:
SuPAR levels in SARS-CoV-2 negative and positive patients varied and amounted to 3.61±1.59 ng/ml vs. 6.45±3.13 ng/ml, respectively (MD = -3.18; 95%CI: -4.71 to -1.66; p<0.001). suPAR levels among non-severe and severe COVID-19 patients were 7.06±2.64 ng/ml and 5.06±3.16 ng/ml (MD = 0.18; 95%CI: -2.48 to 2.83; p=0.90), respectively. Pooled analysis showed that suPAR levels between severe versus critical COVID-19 patients to be 5.59±1.54 ng/ml and 6.49±1.43 ng/ml, respectively (MD = -1.00; 95%CI: -1.31 to -0.70; p<0.001). The suPAR levels between ICU survivors versus non-survivors amounted to 5.82±2.33 ng/ml and 8.43±4.66 ng/ml (MD = -3.59; 95%CI: -6.19 to -1.00; p=0.007). In the case of in-hospital mortality, the mean suPAR level among survivors to hospital discharge was 5.63±1.27 ng/ml, compared to 7.85±2.61 ng/ml for patients who did not survive (MD = -3.58; 95%CI: -5.42 to -1.74; p<0.001).
Conclusions:
SuPAR levels are significantly elevated in severe COVID-19 illness and maybe useful in predicting mortality. Further studies are needed to determine cut-off points and clarify the association of suPAR levels with disease progression. This is of utmost importance given the ongoing pandemic and overburdened health care systems.
In COVID-19, the rapid prediction of the severity of a patient’s condition using modern biomarkers can accelerate the implementation of appropriate therapy, and thus improve the patient’s prognosis.
Material and methods:
A meta-analysis was conducted of data available in the literature on the differences in baseline suPAR blood concentration between patients (1) who tested positive and negative for COVID-19, (2) who had severe and non-severe COVID-19, and (3) COVID-19 survivors and non-survivors.
Results:
SuPAR levels in SARS-CoV-2 negative and positive patients varied and amounted to 3.61±1.59 ng/ml vs. 6.45±3.13 ng/ml, respectively (MD = -3.18; 95%CI: -4.71 to -1.66; p<0.001). suPAR levels among non-severe and severe COVID-19 patients were 7.06±2.64 ng/ml and 5.06±3.16 ng/ml (MD = 0.18; 95%CI: -2.48 to 2.83; p=0.90), respectively. Pooled analysis showed that suPAR levels between severe versus critical COVID-19 patients to be 5.59±1.54 ng/ml and 6.49±1.43 ng/ml, respectively (MD = -1.00; 95%CI: -1.31 to -0.70; p<0.001). The suPAR levels between ICU survivors versus non-survivors amounted to 5.82±2.33 ng/ml and 8.43±4.66 ng/ml (MD = -3.59; 95%CI: -6.19 to -1.00; p=0.007). In the case of in-hospital mortality, the mean suPAR level among survivors to hospital discharge was 5.63±1.27 ng/ml, compared to 7.85±2.61 ng/ml for patients who did not survive (MD = -3.58; 95%CI: -5.42 to -1.74; p<0.001).
Conclusions:
SuPAR levels are significantly elevated in severe COVID-19 illness and maybe useful in predicting mortality. Further studies are needed to determine cut-off points and clarify the association of suPAR levels with disease progression. This is of utmost importance given the ongoing pandemic and overburdened health care systems.
ACKNOWLEDGEMENTS
The study was supported by the ERC Research Net and by
the Polish Society of Disaster Medicine.
REFERENCES (40)
1.
Huang C, Huang L, Wang Y, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021; 397(10270):220–232. https://doi.org/10.1016/S0140-....
2.
Dzieciatkowski T, Szarpak L, Filipiak KJ, et al. COVID-19 challenge for modern medicine. Cardiol J. 2020;27(2):175–183. https://doi.org/10.5603/CJ.a20....
3.
Wolff D, Nee S, Hickey NS, Marschollek M. Risk factors for Covid-19 severity and fatality: a structured literature review. Infection. 2021;49(1):15–28. https://doi.org/10.1007/s15010....
4.
Berzuini C, Hannan C, King A, et al. Value of dynamic clinical and biomarker data for mortality risk prediction in COVID-19: a multicentre retrospective cohort study. BMJ Open. 2020;10(9):e041983. https://doi.org/10.1136/bmjope....
5.
Szarpak L, Ruetzler K, Safiejko K, et al. Lactate dehydrogenase level as a COVID-19 severity marker. Am J Emerg Med. 2021;45:638–639. https://doi.org/10.1016/j.ajem....
6.
Smilowitz NR, Nguy V, Aphinyanaphongs Y, et al. Multiple Biomarker Approach to Risk Stratification in COVID-19. Circulation. 2021;143(13): 1338–1340. https://doi.org/10.1161/CIRCUL....
7.
Szarpak Ł, Nowak B, Kosior D, et al. Cytokines as predictors of COVID-19 severity: evidence from a meta-analysis. Pol Arch Intern Med. 2021;131(1):98–99. https://doi.org/10.20452/pamw.....
8.
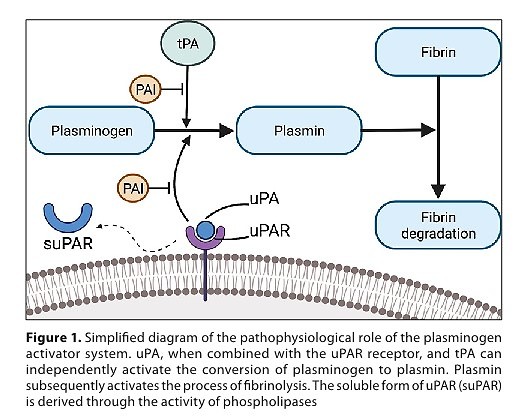
Mahmood N, Mihalcioiu C, Rabbani SA. Multifaceted Role of the Urokinase-Type Plasminogen Activator (uPA) and Its Receptor (uPAR): Diagnostic, Prognostic, and Therapeutic Applications. Front Oncol. 2018;8:24. https://doi.org/10.3389/fonc.2....
9.
Madsen CD, Sidenius N. The interaction between urokinase receptor and vitronectin in cell adhesion and signalling. Eur J Cell Biol. 2008; 87(8–9):617–629. https://doi.org/10.1016/j.ejcb....
10.
Reisinger AC, Niedrist T, Posch F, et al. Soluble urokinase plasminogen activator receptor (suPAR) predicts critical illness and kidney failure in patients admitted to the intensive care unit. Sci Rep. 2021;11(1):17476. https://doi.org/10.1038/s41598....
11.
Schultz M, Rasmussen LJH, Kallemose T, Kjoller E, Lind MN, Ravn L, et al. Availability of suPAR in emergency departments may improve risk stratification: a secondary analysis of the TRIAGE III trial. Scand J Trauma Resusc Emerg Med. 2019;27(1):43. https://doi.org/10.1186/s13049....
12.
Donadello K, Scolletta S, Taccone FS, et al. Soluble urokinase-type plasminogen activator receptor as a prognostic biomarker in critically ill patients. J Crit Care. 2014;29(1):144–149. https://doi.org/10.1016/j.jcrc....
13.
Koller L, Stojkovic S, Richter B, et al. Soluble Urokinase-Type Plasminogen Activator Receptor Improves Risk Prediction in Patients With Chronic Heart Failure. JACC Heart Fail. 2017;5(4):268–277. https://doi.org/10.1016/j.jchf....
14.
Hayek SS, Leaf DE, Samman Tahhan A, et al. Soluble Urokinase Receptor and Acute Kidney Injury. N Engl J Med. 2020;382(5):416–426. https://doi.org/10.1056/NEJMoa....
15.
Chalkias A, Skoulakis A, Papagiannakis N, et al. Circulating suPAR associates with severity and in-hospital progression of COVID-19. Eur J Clin Invest. 2022;52(7):e13794. https://doi.org/10.1111/eci.13....
16.
Napolitano F, Di Spigna G, Vargas M, et al. Soluble Urokinase Receptor as a Promising Marker for Early Prediction of Outcome in COVID-19 Hospitalized Patients. J Clin Med. 2021;10(21):4914. https://doi.org/10.3390/jcm102....
17.
Enocsson H, Idoff C, Gustafsson A, et al. Soluble Urokinase Plasminogen Activator Receptor (suPAR) Independently Predicts Severity and Length of Hospitalisation in Patients With COVID-19. Front Med. 2021;8:791716. https://doi.org/10.3389/fmed.2....
18.
Kerget B, Kerget F, Aksakal A, et al. Evaluation of the relationship between KIM-1 and suPAR levels and clinical severity in COVID-19 patients: A different perspective on suPAR. J Med Virol. 2021;93(9): 5568–5573. https://doi.org/10.1002/jmv.27....
19.
Lo CK, Mertz D, Loeb M. Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014; 14:45. https://doi.org/10.1186/1471-2....
20.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. https://doi.org/10.1186/1471-2....
21.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. https://doi.org/10.1136/bmj.32....
22.
Altintas I, Eugen-Olsen J, Seppälä S, et al. suPAR Cut-Offs for Risk Stratification in Patients With Symptoms of COVID-19. Biomark Insights. 2021;16,11772719211034685. https://doi.org/10.1177/117727....
23.
Genc AB, Aci SY, Dheir H, et al. Super levels and covid-19: Is there a relationship between supar levels and prognosis of covid-19 infection? Acta Medica Mediterranea. 2021;37(5):2503. https://doi.org/10.19193/0393-....
24.
Huang M, Li L, Shen J, Wang Y, et al. Plasma levels of the active form of suPAR are associated with COVID-19 severity. Crit Care. 2020;24(1):704. https://doi.org/10.1186/s13054....
25.
Kakar A, Rani P, Batra T, Hasan R, Choudhury S. A comparative analysis of serial measurements of Soluble Urokinase-type Plasminogen Activator Receptor (suPAR) and C-reactive protein in patients with moderate COVID-19: a single center study from India. medRxiv 2022. https://doi.org/10.1101/2022.0....
26.
Keskinidou C, Vassiliou AG, Zacharis A, et al. Endothelial, Immunothrombotic, and Inflammatory Biomarkers in the Risk of Mortality in Critically Ill COVID-19 Patients: The Role of Dexamethasone. Diagnostics (Basel). 2021;11(7):1249. https://doi.org/10.3390/diagno....
27.
Kyriazopoulou E, Panagopoulos P, Metallidis S, et al. An open label trial of anakinra to prevent respiratory failure in COVID-19. Elife. 2021:10:e66125. https://doi.org/10.7554/eLife.....
28.
Stauning MA, Altintas I, Kallemose T, et al. Soluble Urokinase Plasminogen Activator Receptor as a Decision Marker for Early Discharge of Patients with COVID-19 Symptoms in the Emergency Department. J Emerg Med. 2021;61(3):298–313. https://doi.org/10.1016/j.jeme....
29.
Vassiliou AG, Zacharis A, Vrettou CS, et al. Comparison of the Mortality Prediction Value of Soluble Urokinase Plasminogen Activator Receptor (suPAR) in COVID-19 and Sepsis. Diagnostics (Basel). 2022; 12(5):1261. https://doi.org/10.3390/diagno....
30.
Velissaris D, Lagadinou M, Paraskevas T, et al. Evaluation of Plasma Soluble Urokinase Plasminogen Activator Receptor Levels in Patients With COVID-19 and Non-COVID-19 Pneumonia: An Observational Cohort Study. J Clin Med Res. 2021;13(9):474–478. https://doi.org/10.14740/jocmr....
31.
Infantino M, Morena L, Di Pietro MA, et al. Soluble urokinase Plasminogen Activator Receptor (suPAR) levels are predictive of COVID-19 severity: an Italian experience. Clin Immunol. 2022;242:109091. https://doi.org/10.1016/j.clim....
32.
Böger B, Fachi MM, Vilhena RO, et al. Systematic review with meta-analysis of the accuracy of diagnostic tests for COVID-19. Am J Infect Control. 2021;49(1):21–29. https://doi.org/10.1016/j.ajic....
33.
Rovina N, Akinosoglou K, Eugen-Olsen J, et al. Soluble urokinase plasminogen activator receptor (suPAR) as an early predictor of severe respiratory failure in patients with COVID-19 pneumonia. Crit Care. 2020;24(1):187. https://doi.org/10.1186/s13054....
34.
Kyriazopoulou E, Poulakou G, Milionis H, et al. Early treatment of COVID-19 with anakinra guided by soluble urokinase plasminogen receptor plasma levels: a double-blind, randomized controlled phase 3 trial. Nat Med. 2021;27(10):1752–1760. https://doi.org/10.1038/s41591....
35.
Chen D, Wu X, Yang J, Yu L. Serum plasminogen activator urokinase receptor predicts elevated risk of acute respiratory distress syndrome in patients with sepsis and is positively associated with disease severity, inflammation and mortality. Exp Ther Med. 2019;18(4):2984–2992. https://doi.org/10.3892/etm.20....
36.
Godtfredsen NS, Jorgensen DV, Marsaa K, et al. Soluble urokinase plasminogen activator receptor predicts mortality in exacerbated COPD. Respir Res. 2018;19(1):97. https://doi.org/10.1186/s12931....
37.
Torino C, Pizzini P, Cutrupi S, et al. Soluble Urokinase Plasminogen Activator Receptor (suPAR) and All-Cause and Cardiovascular Mortality in Diverse Hemodialysis Patients. Kidney Int Rep. 2018;3(5):1100–1109. https://doi.org/10.1016/j.ekir....
38.
Wang Y, Wu F, Chen C, Xu L, Lin W, Huang C, et al. Soluble urokinase plasminogen activator receptor is associated with short-term mortality and enhanced reactive oxygen species production in acute-on-chronic liver failure. BMC Gastroenterol. 2021;21(1):429. https://doi.org/10.1186/s12876....
39.
Sando A, Schultz M, Eugen-Olsen J, et al. Soluble urokinase receptor as a predictor of non-cardiac mortality in patients with percutaneous coronary intervention treated ST-segment elevation myocardial infarction. Clin Biochem. 2020;80:8–13. https://doi.org/10.1016/j.clin....
40.
Lin L, Chu H. Quantifying publication bias in meta-analysis. Biometrics. 2018;74(3):785–794. https://doi.org/10.1111/biom.1....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.