Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
On-admission laboratory predictors for developing critical COVID-19 during
hospitalization – a multivariable logistic regression model
1
Department of Rheumatology, Rehabilitation and Internal Diseases, Univeristy of Medical Sciences, Poznań, Poland
2
Rheumatology and Osteoporosis Ward, J. Strus Municipal Hospital, Poznań, Poland
Corresponding author
Wiktor Schmidt
Department of Rheumatology, Rehabilitation and Internal Diseases, Poznan Univeristy of Medical Sciences, Poland
Department of Rheumatology, Rehabilitation and Internal Diseases, Poznan Univeristy of Medical Sciences, Poland
Ann Agric Environ Med. 2022;29(2):274-280
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Recognition of patients with COVID-19 who will progress clinically and need respiratory support remains challenging. The aim of the study was to identify abnormalities in on-admission laboratory results that can precede progression from moderate or severe to critical COVID-19.
Material and methods:
Laboratory data analyzed of 190 patients admitted with moderate or severe COVID-19 to our ward. Laboratory results taken into analysis were obtained during the first 48 hours of hospitalization. Multivariate logistic regression was performed using risk factors obtained in the univariate analysis as dependent variables.
Results:
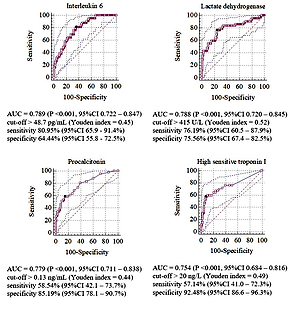
42 patients were identified who developed critical COVID-19. In univariate analysis, 22 laboratory risk factors were detected that were used in logistic regression and in building model with following predictors: high-sensitive troponin I concentration (hs-TnI) >26 ng/mL (OR 13.45; 95%CI 3.28–55.11; P 15 (OR 5.67; 95%CI 1.97–16.36, P 50 pg/mL (OR 5.52; 95%CI 1.86–16.37; P = 0.001), fasting glycaemia >6.8 mmol/L (OR 4.74; 95%CI 1.65–13.66; P = 0.002), immature neutrophils count >0.06/µL (OR 4.06; 95%CI 1.35–12.2; P = 0.012) and urine protein concentration >500 mg/L (OR 2.94; 95%CI 1.04–8.31; P = 0.043).
Conclusions:
The most significant risk factors of developing critical COVID-19 during hospitalization are: elevated hs-TnI, IL-6, and glucose serum concentrations, increased immature neutrophil count, neutrophils to monocytes ratio, and proteinuria during the first 48 hours after admission. The model built with these predictors achieved better predictive performance than any other univariately analysed laboratory markers in predicting the critical development COVID-19.
Recognition of patients with COVID-19 who will progress clinically and need respiratory support remains challenging. The aim of the study was to identify abnormalities in on-admission laboratory results that can precede progression from moderate or severe to critical COVID-19.
Material and methods:
Laboratory data analyzed of 190 patients admitted with moderate or severe COVID-19 to our ward. Laboratory results taken into analysis were obtained during the first 48 hours of hospitalization. Multivariate logistic regression was performed using risk factors obtained in the univariate analysis as dependent variables.
Results:
42 patients were identified who developed critical COVID-19. In univariate analysis, 22 laboratory risk factors were detected that were used in logistic regression and in building model with following predictors: high-sensitive troponin I concentration (hs-TnI) >26 ng/mL (OR 13.45; 95%CI 3.28–55.11; P 15 (OR 5.67; 95%CI 1.97–16.36, P 50 pg/mL (OR 5.52; 95%CI 1.86–16.37; P = 0.001), fasting glycaemia >6.8 mmol/L (OR 4.74; 95%CI 1.65–13.66; P = 0.002), immature neutrophils count >0.06/µL (OR 4.06; 95%CI 1.35–12.2; P = 0.012) and urine protein concentration >500 mg/L (OR 2.94; 95%CI 1.04–8.31; P = 0.043).
Conclusions:
The most significant risk factors of developing critical COVID-19 during hospitalization are: elevated hs-TnI, IL-6, and glucose serum concentrations, increased immature neutrophil count, neutrophils to monocytes ratio, and proteinuria during the first 48 hours after admission. The model built with these predictors achieved better predictive performance than any other univariately analysed laboratory markers in predicting the critical development COVID-19.
REFERENCES (42)
1.
Zhu N, Zhang D, Wang W, et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020; 382(8): 727–733. doi: 10.1056/NEJMoa2001017.
2.
Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The Lancet. 2020; 395(10223): 507–513. doi: 10.1016/S0140-6736(20)30211-7.
3.
Brodin P. Immune determinants of COVID-19 disease presentation and severity. Nat Med. 2021; 27(1): 28–33. doi: 10.1038/s41591-020-01202-8.
4.
Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020; 323(13): 1239–1242. doi: 10.1001/jama.2020.2648.
5.
Gandhi RT, Lynch JB, Del Rio C. Mild or Moderate Covid-19. N Engl J Med. 2020; 383(18): 1757–1766. doi: 10.1056/NEJMcp2009249.
6.
Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N Engl J Med. 2020; 383(25): 2451–2460. doi: 10.1056/NEJMcp2009575.
7.
Del Sole F, Farcomeni A, Loffredo L, et al. Features of severe COVID-19: A systematic review and meta-analysis. Eur J Clin Invest. 2020; 50(10): e13378. doi: 10.1111/eci.13378.
8.
Wynants L, Van Calster B, Collins GS, et al. Prediction models for diagnosis and prognosis of covid-19 infection: systematic review and critical appraisal. BMJ. 2020; 369: m1328. doi: 10.1136/bmj.m1328.
9.
WHO Working Group on the Clinical Characterisation and Management of COVID-19 infection. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis. 2020; 20(8): e192-e197. doi: 10.1016/S1473-3099(20)30483-7.
10.
A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis. 2020; 20(8): e192-e197. doi: 10.1016/S1473-3099(20)30483-7.
11.
Wettstein RB, Shelledy DC, Peters JI. Delivered oxygen concentrations using low-flow and high-flow nasal cannulas. Respir Care. 2005; 50(5): 604–609.
12.
Subbe CP, Kruger M, Rutherford P, et al. Validation of a modified Early Warning Score in medical admissions. QJM Mon J Assoc Physicians. 2001; 94(10): 521–526. doi: 10.1093/qjmed/94.10.521.
13.
Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of Clinical Criteria for Sepsis: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016; 315(8): 762–774. doi: 10.1001/jama.2016.0288.
14.
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987; 40(5): 373–383. doi: 10.1016/0021-9681(87)90171-8.
15.
Chang YC, Yu CJ, Chang SC, et al. Pulmonary sequelae in convalescent patients after severe acute respiratory syndrome: evaluation with thin-section CT. Radiology. 2005; 236(3): 1067–1075. doi: 10.1148/radiol.2363040958.
16.
Chang WT, Toh HS, Liao CT, et al. Cardiac Involvement of COVID-19: A Comprehensive Review. Am J Med Sci. 2021; 361(1): 14–22. doi: 10.1016/j.amjms.2020.10.002.
17.
Piccioni A, Brigida M, Loria V, et al. Role of troponin in COVID-19 pandemic: a review of literature. Eur Rev Med Pharmacol Sci. 2020; 24(19): 10293–10300. doi: 10.26355/eurrev_202010_23254.
18.
Pourbagheri-Sigaroodi A, Bashash D, Fateh F, et al. Laboratory findings in COVID-19 diagnosis and prognosis. Clin Chim Acta Int J Clin Chem. 2020; 510: 475–482. doi: 10.1016/j.cca.2020.08.019.
19.
Hemmat N, Derakhshani A, Bannazadeh Baghi H, et al. Neutrophils, Crucial, or Harmful Immune Cells Involved in Coronavirus Infection: A Bioinformatics Study. Front Genet. 2020; 11: 641. doi: 10.3389/fgene.2020.00641.
20.
Gatti A, Radrizzani D, Viganò P, et al. Decrease of Non-Classical and Intermediate Monocyte Subsets in Severe Acute SARS-CoV-2 Infection. Cytom Part J Int Soc Anal Cytol. 2020; 97(9): 887–890. doi: 10.1002/cyto.a.24188.
21.
McGonagle D, Sharif K, O’Regan A, et al. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun Rev. 2020; 19(6): 102537. doi: 10.1016/j.autrev.2020.102537.
22.
Coomes EA, Haghbayan H. Interleukin-6 in Covid-19: A systematic review and meta-analysis. Rev Med Virol. 2020; 30(6): 1–9. doi: 10.1002/rmv.2141.
23.
Liu F, Li L, Xu M, et al. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol Off Publ Pan Am Soc Clin Virol. 2020; 127: 104370. doi: 10.1016/j.jcv.2020.104370.
24.
Lim S, Bae JH, Kwon HS, et al. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol. 2021; 17(1): 11–30. doi: 10.1038/s41574-020-00435-4.
25.
Ejaz H, Alsrhani A, Zafar A, et al. COVID-19 and comorbidities: Deleterious impact on infected patients. J Infect Public Health. 2020; 13(12): 1833–1839. doi: 10.1016/j.jiph.2020.07.014.
26.
Wu ZH, Tang Y, Cheng Q. Diabetes increases the mortality of patients with COVID-19: a meta-analysis. Acta Diabetol. 2021; 58(2): 139–144. doi: 10.1007/s00592-020-01546-0.
27.
Ronit A, Berg RMG, Bay JT, et al. Compartmental immunophenotyping in COVID-19 ARDS: A case series. J Allergy Clin Immunol. 2021; 147(1): 81–91. doi: 10.1016/j.jaci.2020.09.009.
28.
Schulte-Schrepping J, Reusch N, Paclik D, et al. Severe COVID-19 Is Marked by a Dysregulated Myeloid Cell Compartment. Cell. 2020; 182(6): 1419–1440.e23. doi: 10.1016/j.cell.2020.08.001.
29.
Perico L, Benigni A, Casiraghi F, Ng LFP, et al. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat Rev Nephrol. 2021; 17(1): 46–64. doi: 10.1038/s41581-020-00357-4.
30.
Pei G, Zhang Z, Peng J, et al. Renal Involvement and Early Prognosis in Patients with COVID-19 Pneumonia. J Am Soc Nephrol JASN. 2020; 31(6): 1157–1165. doi: 10.1681/ASN.2020030276.
31.
Canovi S, Besutti G, Bonelli E, et al. The association between clinical laboratory data and chest CT findings explains disease severity in a large Italian cohort of COVID-19 patients. BMC Infect Dis. 2021; 21(1): 157. doi: 10.1186/s12879-021-05855-9.
32.
Zhang B, Zhang J, Chen H, et al. Novel coronavirus disease 2019 (COVID-19): relationship between chest CT scores and laboratory parameters. Eur J Nucl Med Mol Imaging. 2020; 47(9): 2083–2089. doi: 10.1007/s00259-020-04854-3.
33.
Zhou M, Dong C, Li C, et al. Longitudinal changes in COVID-19 clinical measures and correlation with the extent of CT lung abnormalities. Int J Med Sci. 2021; 18(5): 1277–1284. doi: 10.7150/ijms.51279.
34.
Francone M, Iafrate F, Masci GM, et al. Chest CT score in COVID-19 patients: correlation with disease severity and short-term prognosis. Eur Radiol. 2020; 30(12): 6808–6817. doi: 10.1007/s00330-020-07033-y.
35.
Figueira Gonçalves JM, Hernández Pérez JM, Acosta Sorensen M, et al. Biomarkers of acute respiratory distress syndrome in adults hospitalised for severe SARS-CoV-2 infection in Tenerife Island, Spain. BMC Res Notes. 2020; 13: 555. doi: 10.1186/s13104-020-05402-w.
36.
Macias-Muñoz L, Wijngaard R, González-de la Presa B, et al. Value of clinical laboratory test for early prediction of mortality in patients with COVID-19: the BGM score. J Circ Biomark. 2021; 10: 1–8. doi: 10.33393/jcb.2021.2194.
37.
Mehta AA, Haridas N, Belgundi P, et al. A systematic review of clinical and laboratory parameters associated with increased severity among COVID-19 patients. Diabetes Metab Syndr. 2021; 15(2): 535–541. doi: 10.1016/j.dsx.2021.02.020.
38.
Nijman G, Wientjes M, Ramjith J, et al. Risk factors for in-hospital mortality in laboratory-confirmed COVID-19 patients in the Netherlands: A competing risk survival analysis. PloS One. 2021; 16(3): e0249231. doi: 10.1371/journal.pone.0249231.
39.
Statsenko Y, Al Zahmi F, Habuza T, et al. Prediction of COVID-19 severity using laboratory findings on admission: informative values, thresholds, ML model performance. BMJ Open. 2021; 11(2): e044500. doi: 10.1136/bmjopen-2020-044500.
40.
Bennouar S, Bachir Cherif A, Kessira A, et al. Development and validation of a laboratory risk score for the early prediction of COVID-19 severity and in-hospital mortality. Intensive Crit Care Nurs. Published online January 9, 2021:103012. doi: 10.1016/j.iccn.2021.103012.
41.
Leszczyński P. COVID-19: a short message to rheumatologists. Reumatologia. 2020; 58(3): 130–133. doi: 10.5114/reum.2020.96685.
42.
Kanecki K, Nitsch-Osuch A, Goryński P, et al. Hospitalizations for COVID-19 in Poland: a study based on data from a national hospital register. Pol Arch Intern Med. 2021; 131(6): 535–540. doi: 10.20452/pamw.15946.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.