Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Modulation of locomotor training in a mouse model with Bcl-2 gene deletion after sciatic nerve crush injury
1
Department of Sports Medicine and Physiology of Physical Exercise, Faculty of Health Sciences, Silesian Medical University, Katowice, Poland
2
Department of Rehabilitation, Faculty of Health Sciences, Silesian Medical University, Katowice, Poland
3
Department of Experimental Medicine, Faculty of Medical Sciences, Medical University of Silesia, Katowice, Poland
Corresponding author
Natalia Białoń
Department of Sports Medicine and Physiology of Physical Exercise, Faculty of Health Sciences in Katowice, Silesian Medical University of Katowice, Poland
Department of Sports Medicine and Physiology of Physical Exercise, Faculty of Health Sciences in Katowice, Silesian Medical University of Katowice, Poland
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Peripheral nerve injuries are often a traumatic event that requires long-term rehabilitation-based treatment to regain the best possible mobility. One of the promising approaches to enhance recovery involves the use of locomotor training, which may influence neuroplasticity and functional regeneration. The aim of the study is to investigate the effect of forced treadmill training on locomotor functional recovery following sciatic nerve crush in Bcl-2 deficient mice. It is based on an evaluation of the results of a locomotor analysis of mice with an injured sciatic nerve, taking into account specific gait parameters using the Noldus CatWalk device.
Material and methods:
The animals trained on the treadmill for 30 minutes a day,5 days a week for 4 weeks after a crush injury of the sciatic nerve, at a speed (20 m / min), without rest, at 0 degrees and 30 degrees. The present research indicates that the bcl 2 gene plays an important role in regulating neuronal loss following axon injury. Particular attention is drawn to the fact that the functional recovery of mice lacking this gene is weaker than that of wild-type mice.
Results:
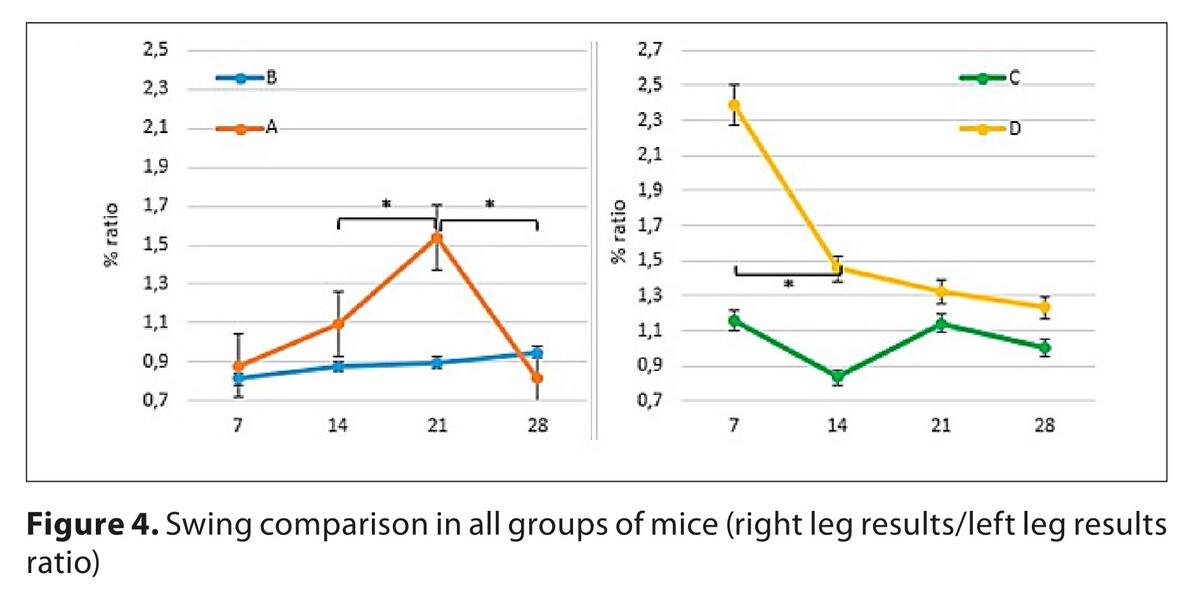
The differences observed in the recovery time of function in individual parameters of the treadmill alignment also reflect the dynamic changes taking place in the regenerating nerve. Mice lacking the bcl-2 gene regain their lost functions less clearly.
Conclusions:
Introduction and Objective. Peripheral nerve injuries are often a traumatic event that requires long-term rehabilitation- based treatment to regain the best possible mobility. One of the promising approaches to enhance recovery involves the use of locomotor training, which may influence neuroplasticity and functional regeneration. The aim of the study is to investigate the effect of forced treadmill training on locomotor functional recovery following sciatic nerve crush in Bcl-2 deficient mice. It is based on an evaluation of the results of a locomotor analysis of mice with an injured sciatic nerve, taking into account specific gait parameters using the Noldus CatWalk device. Materials and Method. The animals trained on the treadmill for 30 minutes a day,5 days a week for 4 weeks after a crush injury of the sciatic nerve, at a speed (20 m / min), without rest, at 0 degrees and 30 degrees. The present research indicates that the bcl 2 gene plays an important role in regulating neuronal loss following axon injury. Particular attention is drawn to the fact that the functional recovery of mice lacking this gene is weaker than that of wild-type mice. Results. The differences observed in the recovery time of function in individual parameters of the treadmill alignment also reflect the dynamic changes taking place in the regenerating nerve. Mice lacking the bcl-2 gene regain their lost functions less clearly. It is worth emphasizing the necessity to use intensified training. The rapid functional recovery in wild-type mice training at different levels thus demonstrates the importance of the presence of the bcl-2 gene, which clearly plays a decisive role here.t is worth emphasizing the necessity to use intensified training. The rapid functional recovery in wild-type mice training at different levels thus demonstrates the importance of the presence of the bcl-2 gene, which clearly plays a decisive role here.
Peripheral nerve injuries are often a traumatic event that requires long-term rehabilitation-based treatment to regain the best possible mobility. One of the promising approaches to enhance recovery involves the use of locomotor training, which may influence neuroplasticity and functional regeneration. The aim of the study is to investigate the effect of forced treadmill training on locomotor functional recovery following sciatic nerve crush in Bcl-2 deficient mice. It is based on an evaluation of the results of a locomotor analysis of mice with an injured sciatic nerve, taking into account specific gait parameters using the Noldus CatWalk device.
Material and methods:
The animals trained on the treadmill for 30 minutes a day,5 days a week for 4 weeks after a crush injury of the sciatic nerve, at a speed (20 m / min), without rest, at 0 degrees and 30 degrees. The present research indicates that the bcl 2 gene plays an important role in regulating neuronal loss following axon injury. Particular attention is drawn to the fact that the functional recovery of mice lacking this gene is weaker than that of wild-type mice.
Results:
The differences observed in the recovery time of function in individual parameters of the treadmill alignment also reflect the dynamic changes taking place in the regenerating nerve. Mice lacking the bcl-2 gene regain their lost functions less clearly.
Conclusions:
Introduction and Objective. Peripheral nerve injuries are often a traumatic event that requires long-term rehabilitation- based treatment to regain the best possible mobility. One of the promising approaches to enhance recovery involves the use of locomotor training, which may influence neuroplasticity and functional regeneration. The aim of the study is to investigate the effect of forced treadmill training on locomotor functional recovery following sciatic nerve crush in Bcl-2 deficient mice. It is based on an evaluation of the results of a locomotor analysis of mice with an injured sciatic nerve, taking into account specific gait parameters using the Noldus CatWalk device. Materials and Method. The animals trained on the treadmill for 30 minutes a day,5 days a week for 4 weeks after a crush injury of the sciatic nerve, at a speed (20 m / min), without rest, at 0 degrees and 30 degrees. The present research indicates that the bcl 2 gene plays an important role in regulating neuronal loss following axon injury. Particular attention is drawn to the fact that the functional recovery of mice lacking this gene is weaker than that of wild-type mice. Results. The differences observed in the recovery time of function in individual parameters of the treadmill alignment also reflect the dynamic changes taking place in the regenerating nerve. Mice lacking the bcl-2 gene regain their lost functions less clearly. It is worth emphasizing the necessity to use intensified training. The rapid functional recovery in wild-type mice training at different levels thus demonstrates the importance of the presence of the bcl-2 gene, which clearly plays a decisive role here.t is worth emphasizing the necessity to use intensified training. The rapid functional recovery in wild-type mice training at different levels thus demonstrates the importance of the presence of the bcl-2 gene, which clearly plays a decisive role here.
REFERENCES (25)
1.
Opferman JT, Kothari A. Anti-apoptotic BCL-2 family members in development. Cell Death Differ. 2018 Jan;25(1):37–45. doi:10.1038/cdd.2017.170. Epub 2017 Nov 3. PMID: 29099482; PMCID: PMC5729530.
2.
Naderer T, Fulcher MC. Targeting apoptosis pathways in infections. J Leukoc Biol. 2018 Feb;103(2):275–285. doi:10.1189/JLB.4MR0717-286R. PMID: 29372933.
3.
Ahmed A, Tait SWG. Molecular Cell Biology of Apoptosis in Health and Disease. Adv Exp Med Biol. 2025;1481:1–28. doi:10.1007/978-3-031-92785-0_1. PMID: 41004078.
4.
Bock FJ, Riley JS. When cell death goes wrong: inflammatory outcomes of failed apoptosis and mitotic cell death. Cell Death Differ. 2023 Feb;30(2):293–303. doi:10.1038/s41418-022-01082-0. Epub 2022 Nov 14. PMID: 36376381; PMCID: PMC9661468.
5.
Wang HY, Zhu CH, Liu DS, et al. Rehabilitation training improves cognitive disorder after cerebrovascular accident by improving BDNF Bcl-2 and Bax expressions in regulating the JMK pathway.EurRev Med Pharmacol Sci. 2021 May;25(10):3807–3821. doi:10.26355/eurrev_202105_25949. PMID: 34109590.
6.
Ruegsegger GN, Booth FW. Health Benefits of Exercise. Cold Spring Harb Perspect Med. 2018 Jul 2;8(7):a029694. doi:10.1101/cshperspect.a029694. PMID: 28507196; PMCID: PMC6027933.
7.
Yamakawa M, Santosa SM, Chawla N, et al. Transgenic models for investigating the nervous system: Currently available neurofluorescent reporters and potential neuronal markers. Biochim Biophys Acta Gen Subj. 2020 Jul;1864(7):129595. doi:10.1016/j.bbagen.2020.129595. Epub 2020 Mar 12. PMID: 32173376; PMCID: PMC7196036.
8.
Paraschiv P. The Influences of Physical Exercises on the Nervous System. Bulletin of the Polytechnic Institute of Iasi. Machine constructions Section. 2025;70:21–25. 10.2478/bipcm-2024-0012.
9.
Wang W, Shao M, Du W, et al. Impact of exhaustive exercise on autonomic nervous system activity: insights from HRV analysis. Front Physiol. 2024;15:1462082. doi:10.3389/fphys.2024.1462082.
10.
Fletcher B, Phillips R, Faust A, et al. Physical exercise to promote regeneration after peripheral nerve injury in animal models: A systematic review. J Orthop Res. 2024 Jul;42(7):1608–1622. doi:10.1002/jor.25792. Epub 2024 Jan 28. PMID: 38282091.
11.
Armada-da-Silva PA, Pereira C, Amado S, et al. Role of physical exercise for improving posttraumatic nerve regeneration. Int Rev Neurobiol. 2013;109:125–49. doi:10.1016/B978-0-12-420045-6.00006-7. PMID: 24093610.
12.
Fahimi S, Oryan S, Ahmadi R, et al. Downregulation of Bax/Bcl-2 Expression During Apoptosis in the Hippocampus of Diabetic Male Wistar Rats: Ameliorative Effects of Peganum harmala Seed Extract. Iran J Pharm Res. 2023 Feb 1;21(1):e132071. doi:10.5812/ijpr-132071. PMID: 36915407; PMCID: PMC10007996.
13.
Wu W, Wei Z, Wu Z, et al. Exercise training alleviates neuronal apoptosis and re-establishes mitochondrial quality control after cerebral ischemia by increasing SIRT3 expression. Cell Biol Toxicol. 2024 Dec 21;41(1):10. doi:10.1007/s10565-024-09957-3. PMID: 39707047; PMCID: PMC11662049.
14.
Liu S, Li Q, Wang H, et al. Exercise as a promising alternative for sciatic nerve injury pain relief: a meta-analysis. Front Neurol. 2024 Jul 31;15:1424050. doi:10.3389/fneur.2024.1424050. PMID:39144705; PMCID: PMC11323690.
15.
Matias Júnior I, Medeiros P, de Freita RL, et al. Effective Parameters for Gait Analysis in Experimental Models for Evaluating Peripheral Nerve Injuries in Rats. Neurospine. 2019 Jun;16(2):305–316. doi:10.14245/ns.1836080.040. Epub 2019 Jan 4. PMID: 30653907; PMCID: PMC6603843.
16.
Horstman GM, Housley SN, Cope TC. Dysregulation of mechanosensory circuits coordinating the actions of antagonist motor pools following peripheral nerve injury and muscle reinnervation. Exp Neurol. 2019 Aug;318:124–134. doi:10.1016/j.expneurol.2019.04.017. Epub 2019 Apr 27. PMID: 31039333; PMCID: PMC6588415.
17.
Sabatier MJ, English AW. Pathways Mediating Activity-Induced Enhancement of Recovery From Peripheral Nerve Injury. Exerc Sport Sci Rev. 2015 Jul;43(3):163–71. doi:10.1249/JES.0000000000000047. PMID: 25906422; PMCID: PMC4470799.
18.
Bai X, Yao M, Zhu X, et al. Baicalin suppresses interleukin-1β-induced apoptosis, inflammatory response, oxidative stress, and extracellular matrix degradation in human nucleus pulposus cells. Immunopharmacol Immunotoxicol. 2023 Dec;45(4):433–442. doi:10.1080/08923973.2023.2165942. Epub 2023 Jan 19. PMID: 36617937.
19.
Fletcher B, Phillips R, Faust A, et al. Physical exercise to promote regeneration after peripheral nerve injury in animal models: A systematic review. J Orthop Res. 2024 Jul;42(7):1608–162.
20.
Arbat-Plana A, Cobianchi S, Herrando-Grabulosa M, et al. Endogenous modulation of TrkB signaling by treadmill exercise after peripheral nerve injury. Neuroscience. 2017 Jan 6;340:188–200. doi:10.1016/j.neuroscience.2016.10.057. Epub 2016 Oct 29. PMID: 27984178.
21.
Mee-Inta O, Zhao ZW, Kuo YM. Physical Exercise Inhibits Inflammation and Microglial Activation. Cells. 2019 Jul 9;8(7):691. doi:10.3390/cells8070691. PMID: 31324021; PMCID: PMC6678635.
22.
Han Z, Zhao H, Tao Z, et al. TOPK Promotes Microglia/Macrophage Polarization towards M2 Phenotype via Inhibition of HDAC1 and HDAC2 Activity after Transient Cerebral Ischemia. Aging Dis. 2018 Apr 1;9(2):235–248. doi:10.14336/AD.2017.0328. PMID: 29896413; PMCID: PMC5963345.
23.
Luo L, Liu M, Xie H, et al. High-Intensity Interval Training Improves Physical Function, Prevents Muscle Loss, and Modulates Macrophage-Mediated Inflammation in Skeletal Muscle of Cerebral Ischemic Mice. Mediators Inflamm. 2021 Nov 20;2021:1849428. doi:10.1155/2021/1849428. PMID: 34845407; PMCID: PMC8627337.
24.
Mota BC, Kelly ÁM. Exercise alters LPS-induced glial activation in the mouse brain. Neuronal Signal. 2020 Dec 2;4(4):NS20200003. doi:10.1042/NS20200003. PMID: 33304620; PMCID: PMC7711064.
25.
Strohm AO, Majewska AK. Physical exercise regulates microglia in health and disease. Front Neurosci. 2024 Jun 7;18:1420322. doi:10.3389/fnins.2024.1420322. PMID: 38911597; PMCID: PMC11192042.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.