Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
The potential of Tanshinone IIA in affecting miR-499-5p/PDCD4 axis in diabetic foot-related endothelial cells
1
Institute of Basic Research in Clinical Medicine, China Academy of Chinese Medical Sciences, China
2
Pharmacy, Langfang Second People’s Hospital, China
3
Traditional Chinese Medicine Dermatology, Hai’an Hospital of Traditional Chinese Medicine, China
4
Hai’an Cao Yongquan Dermatology Clinic, China
5
Endocrinology, The Affiliated Hospital of Xuzhou Medical University, China
These authors had equal contribution to this work
Corresponding author
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Diabetic foot wound healing is a significant challenge for diabetic patients. Tanshinone IIA (Tan IIA) is widely used in treating diabetes and related complications, but its role in diabetic foot wound healing remains unclear. The aim of the study is to investigate the mechanism of Tan IIA in diabetic foot wound healing
Material and methods:
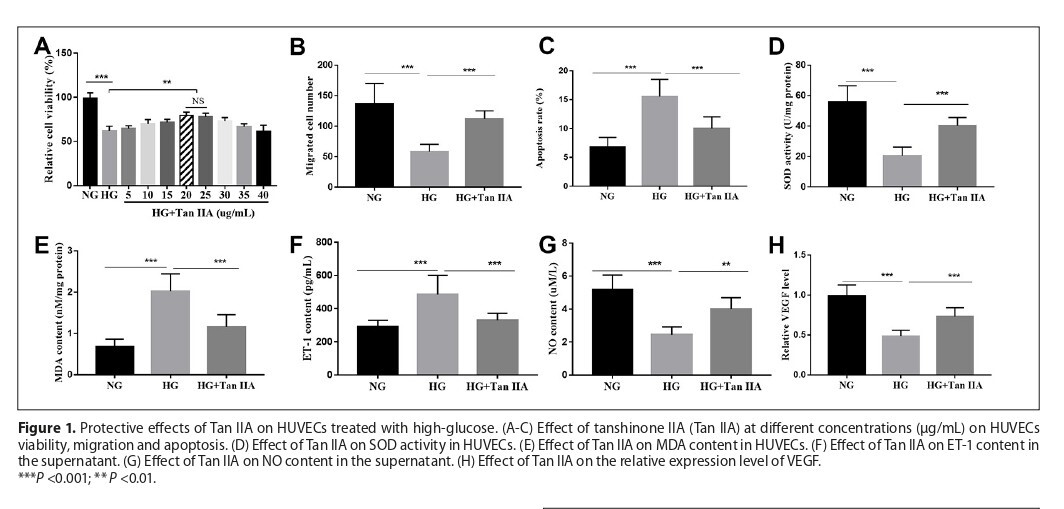
Human umbilical vein endothelial cells (HUVECs) cultured in high-glucose medium were used to simulate diabetic pathological conditions. Cell viability and migration were detected by MTT and transwell assays while Annexin V-FITC/PI was used for apoptosis analysis. Measurement of nitric oxide (NO) and Endothelin-1 (ET-1) were performed by ELISA. The expression of vascular endothelial growth factor (VEGF), miR-499-5p and PDCD4 was calculated by RT-qPCR. Dual-luciferase reporter assay was used to validate the interaction of miR-499-5p and PDCD4.
Results:
Tan IIA protected high-glucose-treated HUVECs by enhancing cell viability and migration, reducing apoptosis and oxidative stress, and improving endothelial function. Similar effects were observed when miR-499-5p was upregulated. Knockdown of miR-499-5p abolished the protective effects of Tan IIA. PDCD4 was identified as a direct target of miR-499-5p.
Conclusions:
Tanshinone IIA protects high glucose-induced HUVECs by enhancing viability, migration, and endothelial function, while reducing apoptosis and oxidative stress, via the miR-499-5p/PDCD4 axis.These in vitro findings support its potential in diabetic foot wound healing, pending in vivo validation.
Diabetic foot wound healing is a significant challenge for diabetic patients. Tanshinone IIA (Tan IIA) is widely used in treating diabetes and related complications, but its role in diabetic foot wound healing remains unclear. The aim of the study is to investigate the mechanism of Tan IIA in diabetic foot wound healing
Material and methods:
Human umbilical vein endothelial cells (HUVECs) cultured in high-glucose medium were used to simulate diabetic pathological conditions. Cell viability and migration were detected by MTT and transwell assays while Annexin V-FITC/PI was used for apoptosis analysis. Measurement of nitric oxide (NO) and Endothelin-1 (ET-1) were performed by ELISA. The expression of vascular endothelial growth factor (VEGF), miR-499-5p and PDCD4 was calculated by RT-qPCR. Dual-luciferase reporter assay was used to validate the interaction of miR-499-5p and PDCD4.
Results:
Tan IIA protected high-glucose-treated HUVECs by enhancing cell viability and migration, reducing apoptosis and oxidative stress, and improving endothelial function. Similar effects were observed when miR-499-5p was upregulated. Knockdown of miR-499-5p abolished the protective effects of Tan IIA. PDCD4 was identified as a direct target of miR-499-5p.
Conclusions:
Tanshinone IIA protects high glucose-induced HUVECs by enhancing viability, migration, and endothelial function, while reducing apoptosis and oxidative stress, via the miR-499-5p/PDCD4 axis.These in vitro findings support its potential in diabetic foot wound healing, pending in vivo validation.
FUNDING
The study was funded by the National Major S & T Special
Project for Significant New Drugs Development (Special
Project No. 2017ZX09301059).
REFERENCES (37)
1.
Korzon-Burakowska A, Dziemidok P. Diabetic foot – the need for comprehensive multidisciplinary approach. Ann Agric Environ Med. 2011;18(2):314–7.
2.
Sanchez ML, Valdez H, Conde M, et al. Polymers and Bioactive Compounds with a Macrophage Modulation Effect for the Rational Design of Hydrogels for Skin Regeneration. Pharmaceutics. 2023;15(6).
3.
Volmer-Thole M, Lobmann R. Neuropathy and Diabetic Foot Syndrome. Int J Mol Sci. 2016;17(6).
4.
Bus SA, Lavery LA, Monteiro-Soares M, et al. Guidelines on the prevention of foot ulcers in persons with diabetes (IWGDF 2019 update). Diabetes Metab Res Rev. 2020;36 Suppl 1:e3269.
5.
Everett E, Mathioudakis N. Update on management of diabetic foot ulcers. Ann N Y Acad Sci. 2018;1411(1):153–65.
6.
Liu Y, Mo J, Liang F, et al. Pien-tze-huang promotes wound healing in streptozotocin-induced diabetes models associated with improving oxidative stress via the Nrf2/ARE pathway. Front Pharmacol. 2023;14:1062664.
7.
Xu Z, Cai K, Su SL, et al. Salvianolic acid B and tanshinone IIA synergistically improve early diabetic nephropathy through regulating PI3K/Akt/NF-kappaB signaling pathway. J Ethnopharmacol. 2024;319(Pt 3):117356.
8.
Wu Q, Guan YB, Zhang KJ, et al. Tanshinone IIA mediates protection from diabetes kidney disease by inhibiting oxidative stress induced pyroptosis. J Ethnopharmacol. 2023;316:116667.
9.
Chen L, He W, Peng B, et al. Sodium Tanshinone IIA sulfonate improves post-ischemic angiogenesis in hyperglycemia. Biochem Biophys Res Commun. 2019;520(3):580–5.
10.
Zhu J, Chen H, Guo J, et al. Sodium Tanshinone IIA Sulfonate Inhibits Vascular Endothelial Cell Pyroptosis via the AMPK Signaling Pathway in Atherosclerosis. J Inflamm Res. 2022;15:6293–306.
11.
Yan C, Chen J, Wang C, et al. Milk exosomes-mediated miR-31-5p delivery accelerates diabetic wound healing through promoting angiogenesis. Drug Deliv. 2022;29(1):214–28.
12.
Ozdemir D, Feinberg MW. MicroRNAs in diabetic wound healing: Pathophysiology and therapeutic opportunities. Trends Cardiovasc Med. 2019;29(3):131–7.
13.
Sun S, Ruan Y, Yan M, et al. Ferulic Acid Alleviates Oxidative Stress-Induced Cardiomyocyte Injury by the Regulation of miR-499-5p/p21 Signal Cascade. Evid Based Complement Alternat Med. 2021;2021:1921457.
14.
Sheng Y, Yang Z, Feng Z, et al. MicroRNA-499-5p promotes vascular smooth muscle cell proliferation and migration via inhibiting SOX6. Physiol Genomics. 2023;55(2):67–74.
15.
Zhao L, Li W, Zhao H. Inhibition of long non-coding RNA TUG1 protects against diabetic cardiomyopathy induced diastolic dysfunction by regulating miR-499-5p. Am J Transl Res. 2020;12(3):718–30.
16.
Zhu H, Chen Z, Ma Z, et al. Tanshinone IIA Protects Endothelial Cells from H₂O₂-Induced Injuries via PXR Activation. Biomolecules & therapeutics. 2017;25(6):599–608.
17.
Johansen OE, Birkeland KI, Jorgensen AP, et al. Diabetic foot ulcer
burden may be modified by high-dose atorvastatin: A 6-month
randomized controlled pilot trial. J Diabetes. 2009;1(3):182–7.
18.
Li Y, Wang X, Chen J, et al. Structural analysis and accelerating wound healing function of a novel galactosylated glycosaminoglycan from the snail Helix lucorum. Carbohydr Polym. 2025;348(Pt B):122900.
19.
Li YH, Xu Q, Xu WH, et al. Mechanisms of protection against diabetesinduced impairment of endothelium-dependent vasorelaxation by Tanshinone IIA. Biochim Biophys Acta. 2015;1850(4):813–23.
20.
Liu B, Liu L, Zang A, et al. Tanshinone IIA inhibits proliferation and induces apoptosis of human nasopharyngeal carcinoma cells via p53-cyclin B1/CDC2. Oncol Lett. 2019;18(3):3317–22.
21.
Nie ZY, Zhao MH, Cheng BQ, et al. Tanshinone IIA regulates human AML cell proliferation, cell cycle, and apoptosis through miR-497-5p/AKT3 axis. Cancer Cell Int. 2020;20:379.
22.
Zhang L, Yang F. Tanshinone IIA improves diabetes-induced renal fibrosis by regulating the miR-34-5p/Notch1 axis. Food Sci Nutr. 2022;10(11):4019–40.
23.
Yang C, Mu Y, Li S, et al. Tanshinone IIA: a Chinese herbal ingredient for the treatment of atherosclerosis. Front Pharmacol. 2023;14:1321880.
24.
Lu TC, Wu YH, Chen WY, Hung YC. Targeting Oxidative Stress and Endothelial Dysfunction Using Tanshinone IIA for the Treatment of Tissue Inflammation and Fibrosis. Oxid Med Cell Longev. 2022;2022:2811789.
25.
Chen L, Guo QH, Chang Y, et al. Tanshinone IIA ameliorated endothelial dysfunction in rats with chronic intermittent hypoxia. Cardiovasc Pathol. 2017;31:47–53.
26.
Qin C, Liu S, Zhou S, et al. Tanshinone IIA promotes vascular normalization and boosts Sorafenib’s anti-hepatoma activity via modulating the PI3K-AKT pathway. Front Pharmacol. 2023;14:1189532.
27.
Han D, Wu X, Liu L, et al. Sodium tanshinone IIA sulfonate protects ARPE-19 cells against oxidative stress by inhibiting autophagy and apoptosis. Sci Rep. 2018;8(1):15137.
28.
Yang JX, Pan YY, Ge JH, et al. Tanshinone II A Attenuates TNF-alpha-Induced Expression of VCAM-1 and ICAM-1 in Endothelial Progenitor Cells by Blocking Activation of NF-kappaB. Cell Physiol Biochem. 2016;40(1–2):195–206.
29.
Wang X, Wu C. Tanshinone IIA improves cardiac function via regulating miR-499-5p dependent angiogenesis in myocardial ischemic mice. Microvasc Res. 2022;143:104399.
30.
Li Y, Lu J, Bao X, et al. MiR-499-5p protects cardiomyocytes against ischaemic injury via anti-apoptosis by targeting PDCD4. Oncotarget. 2016;7(24):35607–17.
31.
Lu K, Chen Q, Li M, et al. Programmed cell death factor 4 (PDCD4), a novel therapy target for metabolic diseases besides cancer. Free Radic Biol Med. 2020;159:150–63.
32.
Merline R, Moreth K, Beckmann J, et al. Signaling by the matrix proteoglycan decorin controls inflammation and cancer through PDCD4 and MicroRNA-21. Sci Signal. 2011;4(199):ra75.
33.
Zhang J, Zhang M, Yang Z, et al. PDCD4 deficiency ameliorates left ventricular remodeling and insulin resistance in a rat model of type 2 diabetic cardiomyopathy. BMJ Open Diabetes Res Care. 2020;8(1).
34.
Li T, Ma Y, Wang M, et al. Platelet-rich plasma plays an antibacterial, anti-inflammatory and cell proliferation-promoting role in an in vitro model for diabetic infected wounds. Infect Drug Resist. 2019;12:297–309.
35.
Pin G, Huanting L, Chengzhan Z, et al. Down-Regulation of PDCD4 Promotes Proliferation, Angiogenesis and Tumorigenesis in Glioma Cells. Front Cell Dev Biol. 2020;8:593685.
36.
Song S, Qiu X. LncRNA miR503HG inhibits epithelial-mesenchymal transition and angiogenesis in hepatocellular carcinoma by enhancing PDCD4 via regulation of miR-15b. Dig Liver Dis. 2021;53(1):107–16.
37.
Zhao SF, Ye YX, Xu JD, et al. Long non-coding RNA KCNQ1OT1 increases the expression of PDCD4 by targeting miR-181a-5p, contributing to cardiomyocyte apoptosis in diabetic cardiomyopathy. Acta Diabetol. 2021;58(9):1251–67.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.