Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
REVIEW PAPER
Evidence for external beam radiotherapy in mediastinal Hodgkin and non-Hodgkin lymphoma – systematic review
1
Healthcare Services Department, Agency for Health Technology Assessment and Tariff System, Warsaw, Poland
2
Chair of Epidemiology and Preventive Medicine, Department of Epidemiology, Faculty of Medicine, Jagiellonian University Medical College, Kraków, Poland
3
Health Policy and Management Department; Institute of Public Health; Faculty of Health Sciences; Jagiellonian University Medical College, Kraków, Poland
4
Department of Nutrition and Drug Research, Institute of Public Health; Faculty of Health Sciences; Jagiellonian University Medical College, Kraków, Poland
Corresponding author
Magdalena Koperny
The Agency for Health Technology Assessment and Tariff System, Healthcare Services Department, Przeskok 2, 00-032, Warszawa, Poland
The Agency for Health Technology Assessment and Tariff System, Healthcare Services Department, Przeskok 2, 00-032, Warszawa, Poland
Ann Agric Environ Med. 2024;31(1):47-56
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Proton beam therapy (PBT) provides the opportunity for a more localized delivery of high energy protons and may reduce the damage to healthy tissues and vital organs. The aim of this review was to assess the effects of proton therapy for patients diagnosed with Hodgkin or non-Hodgkin lymphoma treated with mediastinal irradiation.
Review methods:
A systematic search of EMBASE, MEDLINE via OVID and Cochrane Library was conducted in May 2022 according to PRISMA guidelines to identify relevant data on the efficacy and toxicity of proton beam therapy for patients diagnosed with Hodgkin or non-Hodgkin lymphoma.
Brief description of the state of knowledge:
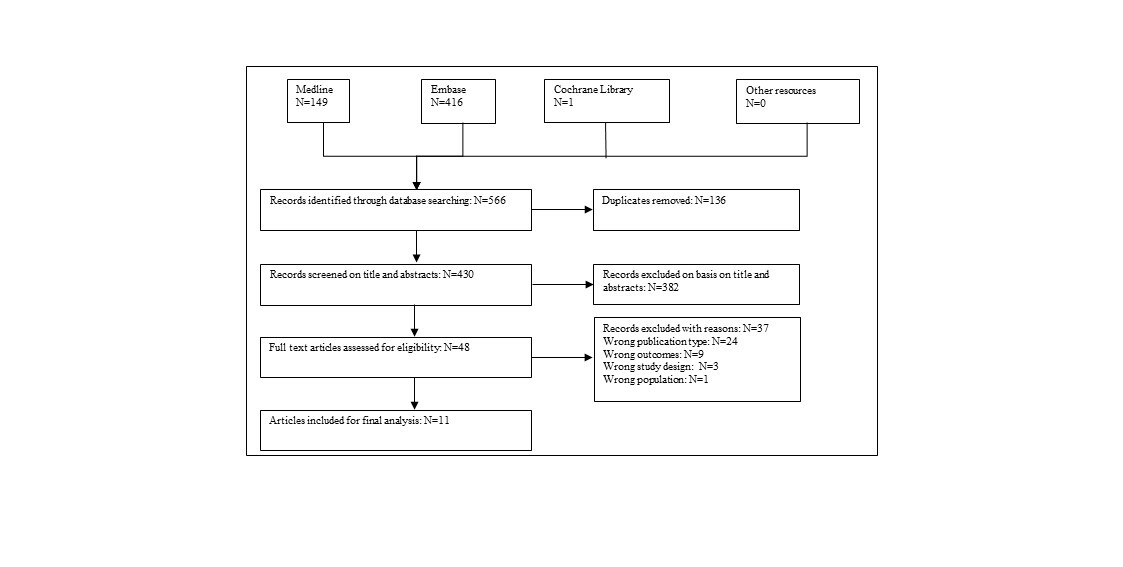
Of 566 screened abstracts (430 after de-duplication) 11 studies with a total of 529 patients were included. All studies were case series published between 2011–2021. Median range of follow-up time was 15–63.6 months. The overall survival (OS) for 2 years varied from 91% – 98% for 5 of the included studies. Three of the included studies had favourable outcomes with 2-year progression-free survival (PFS) ranging from 73% – 94%. Skin reaction, oesophagitis and fatigue were found to be the most common grade 1 and grade 2 toxicities. No acute or late grade 4 and higher toxicities/adverse events were observed.
Summary:
There are data indicating that PBT may to be an effective treatment against mediastinal Hodgkin and non-Hodgkin lymphoma. Because all the studies were case series, the authors of this review have little confidence in the evidence. There remains a need for well-designed randomized controlled trials to inform about the optimal approach to proton irradiation in HL and NHL.
Proton beam therapy (PBT) provides the opportunity for a more localized delivery of high energy protons and may reduce the damage to healthy tissues and vital organs. The aim of this review was to assess the effects of proton therapy for patients diagnosed with Hodgkin or non-Hodgkin lymphoma treated with mediastinal irradiation.
Review methods:
A systematic search of EMBASE, MEDLINE via OVID and Cochrane Library was conducted in May 2022 according to PRISMA guidelines to identify relevant data on the efficacy and toxicity of proton beam therapy for patients diagnosed with Hodgkin or non-Hodgkin lymphoma.
Brief description of the state of knowledge:
Of 566 screened abstracts (430 after de-duplication) 11 studies with a total of 529 patients were included. All studies were case series published between 2011–2021. Median range of follow-up time was 15–63.6 months. The overall survival (OS) for 2 years varied from 91% – 98% for 5 of the included studies. Three of the included studies had favourable outcomes with 2-year progression-free survival (PFS) ranging from 73% – 94%. Skin reaction, oesophagitis and fatigue were found to be the most common grade 1 and grade 2 toxicities. No acute or late grade 4 and higher toxicities/adverse events were observed.
Summary:
There are data indicating that PBT may to be an effective treatment against mediastinal Hodgkin and non-Hodgkin lymphoma. Because all the studies were case series, the authors of this review have little confidence in the evidence. There remains a need for well-designed randomized controlled trials to inform about the optimal approach to proton irradiation in HL and NHL.
REFERENCES (47)
1.
Proton beam therapy. England NHS. Available online: https://www.england.nhs.uk/com... (access: 2022.5.20).
2.
Wrońska A, Ruciński A. Wyzwania w terapii protonowej – jak leczyć nowotwory lepiej? In: Danielewska A, Maciąg M, editors. Nowoczesne technologie XXI w. – przegląd, trendy i badania. 2019. p. 215–229.
3.
Uzasadnienie do rozporządzenia Ministra Zdrowia z dnia 6 czerwca 2016 r. zmieniającego rozporządzenie w sprawie świadczeń gwarantowanych z zakresu leczenia szpitalnego (DzU 2016 poz. 855) Isap.sejm.gov.pl. 2016. Available online: https://isap.sejm.gov.pl/isap.... (access: 2022.5.20).
4.
Hu M, Jiang L, Cui X, Zhang J, Yu J. Proton beam therapy for cancer in the era of precision medicine. J Hematol Oncol. 2018;11(1):136. https://doi.org/10.1186/s13045....
5.
Loap P, Mirandola A, De Marzi L, et al. Current Situation of Proton Therapy for Hodgkin Lymphoma: From Expectations to Evidence. Cancers (Basel). 2021;13(15):3746. https://doi.org/10.3390/cancer....
6.
Kaseb H, Babiker H. Hodgkin Lymphoma. Treasure Island (FL): StatPearls Publishing. Available online: https://www.ncbi.nlm.nih.gov/b... (access: 2022.5.19).
7.
Sapkota S, Shaikh H. Non-Hodgkin Lymphoma England NHS 2022. Available online: https://www.england.nhs.uk/wp-... (access: 2022.5.19).
8.
Cheng J. Ann Arbor staging system Radiology Reference Article Radiopaedia.org 2022. Available online: https://doi.org/10.53347/rID-6... (access: 2022.5.25).
9.
Cheng J. Lugano staging classification Radiology Reference Article Radiopaedia.org 2022. Available online: https://doi.org/10.53347/rID-6... (access: 2022.5.25).
10.
Szymański P. Radioterapia Medycyna Praktyczna 2022. Available online: https://www.mp.pl/pacjent/bada... (access: 2022.5.20).
11.
Hodgkin Lymphoma Treatment Protocol Cancer.org. 2022 Available online: https://www.cancer.org/cancer/... (access: 2022.5.23).
12.
Goitein M, Cox JD. Should randomized clinical trials be required for proton radiotherapy? J Clin Oncol. 2008;26(2):175–176. https://doi.org/10.1200/jco.20....
13.
Tseng YD, Maes SM, Kicska G, et al. Comparative photon and proton dosimetry for patients with mediastinal lymphoma in the era of Monte Carlo treatment planning and variable relative biological effectiveness. Radiat Oncol. 2019;14(1):243. https://doi.org/10.1186/s13014....
14.
Proton Beam Radiation Therapy. Clinical Practice Guideline Albertahealthservices.ca. 2019. Available online: https://www.albertahealthservi... (access: 2022.5.20).
15.
NCCN Guidelines on Proton Therapy. M.incsg. 2018. Available online: http://m.incsg.com/shenjingwai... (access: 2022.5.20).
16.
Proton Beam Radiation Therapy. Guideline Number: MMG113.P. Uhcprovider.com. 2021. Available online: https://www.uhcprovider.com/co... (access: 2022.5.20).
17.
American Imaging Management Inc Aimspecialtyhealth.com. 2022. Available online: https://aimspecialtyhealth.com... (access: 2022.5.20).
18.
Clinical Commissioning Policy Statement Proton Beam Therapy for Head and Neck Cancer in Adults England.nhs.uk. 2019. Available online: https://www.england.nhs.uk/wp-... (access: 2022.5.20).
19.
Clinical Commissioning Policy Statement Proton Beam Therapy for Adult Lymphoma England.nhs.uk. 2019. Available online: https://www.england.nhs.uk/wp-... (access: 2022.5.20).
20.
Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. https://doi.org/10.1136/bmj.l4....
21.
Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. https://doi.org/10.1136/bmj.i4....
22.
Wells G, Wells G, Shea B, et al. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses 2014. Available online: http://www.ohri.ca/programs/cl... (access: 2022.6.09).
23.
Critical Appraisal Tools Jbi.global. 2022. Available online: https://jbi.global/critical-ap... (access: 2022.5.25).
24.
Bates JE, Terezakis S, Morris CG, et al. Comparative Effectiveness of Proton Therapy versus Photon Radiotherapy in Adolescents and Young Adults for Classical Hodgkin Lymphoma. Int J Part Ther. 2021;8(3):21–27. https://doi.org/10.14338/IJPT-....
25.
Dionisi F, Scartoni D, Rombi B, et al. Consolidative active scanning proton therapy for mediastinal lymphoma: selection criteria, treatment implementation and clinical feasibility. Strahlenther Onkol. 2022;198(6):558–565. https://doi.org/10.1007/s00066....
26.
Hoppe BS, Flampouri S, Zaiden R, et al. Involved-node proton therapy in combined modality therapy for Hodgkin lymphoma: results of a phase 2 study. Int J Radiat Oncol Biol Phys. 2014;89(5):1053–1059. https://doi.org/10.1016/j.ijro....
27.
Hoppe BS, Hill-Kayser CE, Tseng YD, et al. Consolidative proton therapy after chemotherapy for patients with Hodgkin lymphoma. Ann Oncol. 2017;28(9):2179–2184. https://doi.org/10.1093/annonc....
28.
König L, Bougatf N, Hörner-Rieber J, et al. Consolidative mediastinal irradiation of malignant lymphoma using active scanning proton beams: clinical outcome and dosimetric comparison. Konsolidierende mediastinale Bestrahlung maligner Lymphome mittels aktivem Rasterscanning mit Protonenstrahlen: Klinische Ergebnisse und dosimetrischer Vergleich. Strahlenther Onkol. 2019;195(7):677–687. https://doi.org/10.1007/s00066....
29.
Li J, Dabaja B, Reed V, et al. Rationale for and preliminary results of proton beam therapy for mediastinal lymphoma. Int J Radiat Oncol Biol Phys. 2011;81(1):167–174. https://doi.org/10.1016/j.ijro....
30.
Loap P, Goudjil F, Dendale R, Kirova Y. Clinical and technical considerations for mediastinal Hodgkin lymphoma protontherapy based on a single-center early experience. Cancer Radiother. 2021;25(8):779–785. https://doi.org/10.1016/j.canr....
31.
Nanda R, Flampouri S, Mendenhall NP, et al. Pulmonary Toxicity Following Proton Therapy for Thoracic Lymphoma. Int J Radiat Oncol Biol Phys. 2017;99(2):494–497. https://doi.org/10.1016/j.ijro....
32.
Tringale KR, Modlin LA, Sine K, Forlenza CJ, Cahlon O, Wolden SL. Vital organ sparing with proton therapy for pediatric Hodgkin lymphoma: Toxicity and outcomes in 50 patients. Radiother Oncol. 2022;168:46–52. https://doi.org/10.1016/j.rado....
33.
Tseng YD, Hoppe BS, Dedeckova K, et al. Risk of Pneumonitis and Outcomes After Mediastinal Proton Therapy for Relapsed/Refractory Lymphoma: A PTCOG and PCG Collaboration. Int J Radiat Oncol Biol Phys. 2021;109(1):220–230. https://doi.org/10.1016/j.ijro....
34.
Wray J, Flampouri S, Slayton W, et al. Proton Therapy for Pediatric Hodgkin Lymphoma. Pediatr Blood Cancer. 2016;63(9):1522–1526. https://doi.org/10.1002/pbc.26....
35.
Delgado-López PD, Corrales-García EM. Role of adjuvant radiotherapy in atypical (WHO grade II) and anaplastic (WHO grade III) meningiomas: a systematic review. Clin Transl Oncol. 2021;23(2):205–221. https://doi.org/10.1007/s12094....
36.
Li JY, Li JW, Jin YC, et al. The Efficacy and Safety of Carbon Ion Radiotherapy for Meningiomas: A Systematic Review and Meta-Analysis. Front Oncol. 2021;11:620534. https://doi.org/10.3389/fonc.2....
37.
Alahmari M, Temel Y. Skull base chordoma treated with proton therapy: A systematic review. Surg Neurol Int. 2019;10:96. https://doi.org/10.25259/SNI-2....
38.
Lee A, Kitpanit S, Chilov M, Langendijk JA, Lu J, Lee NY. A Systematic Review of Proton Therapy for the Management of Nasopharyngeal Cancer. Int J Part Ther. 2021;8(1):119–130. https://doi.org/10.14338/IJPT-....
39.
Lesueur P, Calugaru V, Nauraye C, et al. Proton therapy for treatment of intracranial benign tumors in adults: A systematic review. Cancer Treat Rev. 2019;72:56–64. https://doi.org/10.1016/j.ctrv....
40.
Thurin E, Nyström PW, Smits A, et al. Proton therapy for low-grade gliomas in adults: A systematic review. Clin Neurol Neurosurg. 2018;174:233–238. https://doi.org/10.1016/j.clin....
41.
Hariton E, Locascio JJ. Randomised controlled trials – the gold standard for effectiveness research: Study design: randomised controlled trials. BJOG. 2018;125(13):1716. https://doi.org/10.1111/1471-0....
42.
Mailhot Vega RB, Mohammadi H, Patel SI, et al. Establishing Cost-Effective Allocation of Proton Therapy for Patients With Mediastinal Hodgkin Lymphoma. Int J Radiat Oncol Biol Phys. 2022;112(1):158–166. https://doi.org/10.1016/j.ijro....
43.
Glatstein E, Glick J, Kaiser L, Hahn SM. Should randomized clinical trials be required for proton radiotherapy? An alternative view. J Clin Oncol. 2008;26(15):2438–2439. https://doi.org/10.1200/jco.20....
44.
Baumann BC, Mitra N, Harton JG, et al. Comparative Effectiveness of Proton vs Photon Therapy as Part of Concurrent Chemoradiotherapy for Locally Advanced Cancer. JAMA Oncol. 2020;6(2):237–246. https://doi.org/10.1001/jamaon....
45.
Tian X, Liu K, Hou Y, Cheng J, Zhang J. The evolution of proton beam therapy: Current and future status. Mol Clin Oncol. 2018;8(1):15–21. https://doi.org/10.3892/mco.20....
46.
Schippers JM, Lomax A, Garonna A, Parodi K. Can Technological Improvements Reduce the Cost of Proton Radiation Therapy? Semin Radiat Oncol. 2018;28(2):150–159. https://doi.org/10.1016/j.semr....
47.
Loap P, De Marzi L, Mirandola A, et al. Development and Implementation of Proton Therapy for Hodgkin Lymphoma: Challenges and Perspectives. Cancers (Basel). 2021;13(15):3744. https://doi.org/10.3390/cancer....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.