Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Brain-derived neurotrophic factor and matrix metalloproteinase-9 activity during rehabilitation therapy of schizophrenic patients – environmental pilot study
1
Medical University, Lublin, Poland
2
Department of Women’s Health, Institute of Rural Heath, Lublin, Poland
3
Department of Psychiatry, Faculty of Medicine, Medical University, Warsaw, Poland
Ann Agric Environ Med. 2023;30(2):315-321
KEYWORDS
schizophreniarehabilitationbrain-derived neurotrophic factormatrix metalloproteinase-9neurofeedbackclinical trial
TOPICS
ABSTRACT
Introduction and objective:
The aim of the study was to evaluate the peripheral level of brain-derived neurotrophic factor (BDNF) and matrix metalloproteinase-9 (MMP-9) during rehabilitation therapy, combined with neurofeedback in schizophrenic patients, and to investigate whether these biomarkers are related to psychopathological symptoms, changes in auditory evoked potentials (AEPs), and quantitative EEG (QEEGs) mapping.
Material and methods:
The study involved two groups of patients diagnosed with paranoid schizophrenia in partial remission who participated in a 3-month structured rehabilitation programme combined with neurofeedback (REH group) and a standard support group (CON group). The following parameters were assessed: BDNF and MMP-9 serum levels, AEPs, QEEGs, and psychopathological symptoms (PANSS).
Results:
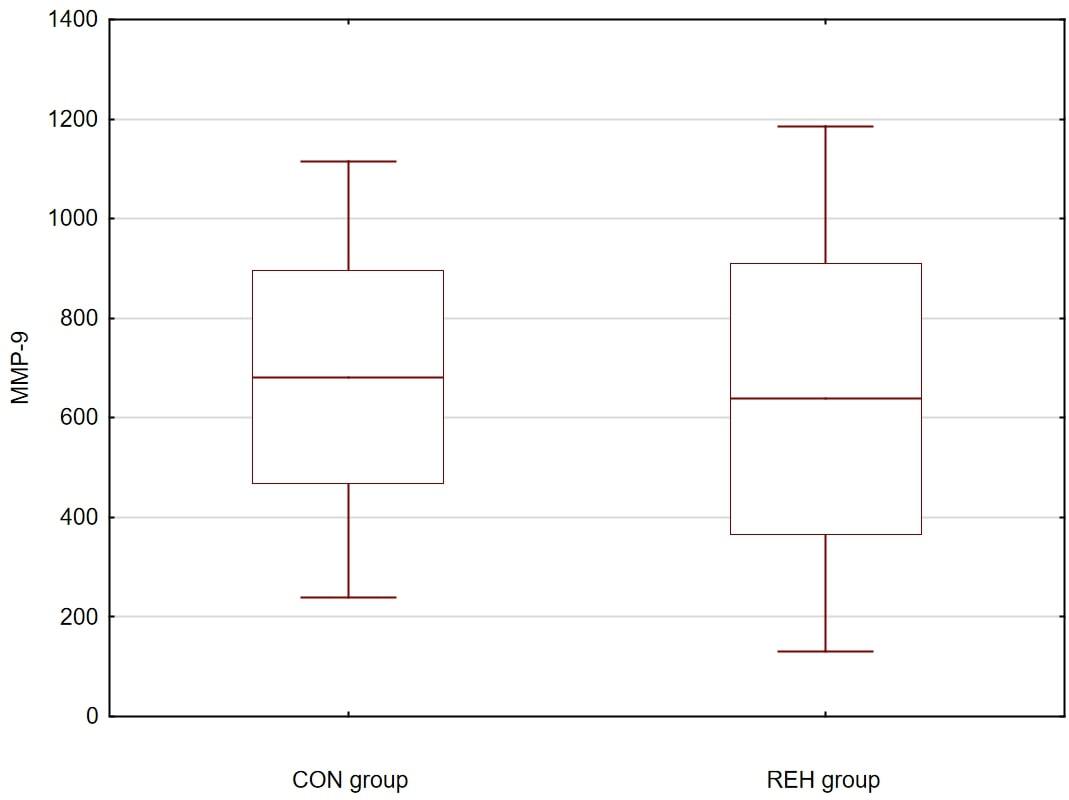
A clinical improvement within the 3-month rehabilitation therapy course was correlated with the increase in BDNF and MMP-9 serum level. Despite the increase in BDNF and MMP-9 during the 3-month rehabilitation therapy, it was not possible to demonstrate any strong and significant correlation between the 2 examined neuropeptides. During the 3-month rehabilitation therapy, the theta waveform share reduction in QEEG, P50 latency reduction and amplitude increase correlated with PANSS Total and MMP-9 results.
Conclusions:
All clinical (PANSS Positive, Negative, General, Total) and biochemical results (BDNF, MMP-9) of the REH group changed significantly over the 3-month period. Positive symptoms improved only in the CON group.
The aim of the study was to evaluate the peripheral level of brain-derived neurotrophic factor (BDNF) and matrix metalloproteinase-9 (MMP-9) during rehabilitation therapy, combined with neurofeedback in schizophrenic patients, and to investigate whether these biomarkers are related to psychopathological symptoms, changes in auditory evoked potentials (AEPs), and quantitative EEG (QEEGs) mapping.
Material and methods:
The study involved two groups of patients diagnosed with paranoid schizophrenia in partial remission who participated in a 3-month structured rehabilitation programme combined with neurofeedback (REH group) and a standard support group (CON group). The following parameters were assessed: BDNF and MMP-9 serum levels, AEPs, QEEGs, and psychopathological symptoms (PANSS).
Results:
A clinical improvement within the 3-month rehabilitation therapy course was correlated with the increase in BDNF and MMP-9 serum level. Despite the increase in BDNF and MMP-9 during the 3-month rehabilitation therapy, it was not possible to demonstrate any strong and significant correlation between the 2 examined neuropeptides. During the 3-month rehabilitation therapy, the theta waveform share reduction in QEEG, P50 latency reduction and amplitude increase correlated with PANSS Total and MMP-9 results.
Conclusions:
All clinical (PANSS Positive, Negative, General, Total) and biochemical results (BDNF, MMP-9) of the REH group changed significantly over the 3-month period. Positive symptoms improved only in the CON group.
ABBREVIATIONS
AEPs – auditory evoked potentials; BDNF – brain-derived neurotrophic factor; CNS – central nervous system; DC – direct
current; EEG – electroencephalogram; GSR – galvanic skin response; ICD – international classification of diseases; MMP-
9 – matrix metalloproteinase-9; NF – neurofeedback; NPS – neuropeptide S; PANSS – positive and negative syndrome
scale; SCL – skin conductance level; SCRs – skin conductance responses; TIMP – tissue inhibitor of metalloproteinease-9;
QEEG – quantitative electroencephalogram
REFERENCES (60)
1.
Rodriguez B, Nani JV, Almeida PGC, Brietzke E, Lee RS, Hayashi MAF. Neuropeptides and oligopeptidases in schizophrenia. Neurosci Biobeh Rev. 2020;108:679–693. doi: https://doi.org/10.1016/j.neub....
2.
Yamamori H, Hashimoto R, Ishima T, et al. Plasma levels of mature brain-derived neurotrophic factor (BDNF) and matrix metalloproteinase-9 (MMP9) in treatment-resistant schizophrenia treated with olanzapine. Neurosci Letter. 2013;556:37–41. doi: https://doi.org/10.1016/j.neul....
3.
Peng S, Li W, Lv L, Zhang Z, Zhan X. BDNF as a biomarker in diagnosis and evaluation of treatment for schizophrenia and depression. Discovery Med. 2018;26(143):127–136.
4.
Markiewicz-Gospodarek A, Markiewicz R, Dobrowolska B, Rahnama M, Łoza B. Relationship of neuropeptide S (NPS) with neurocognitive, clinical, and electrophysiological parameters of patients during structured rehabilitation therapy for schizophrenia. J Clin Med. 2022;11(18):5266. doi: https://doi.org/10.3390/jcm111....
5.
Pan L, Cao Z, Chen L, Qian M, Yan Y. Association of BDNF and MMP-9 single-nucleotide polymorphisms with the clinical phenotype of schizophrenia. Front Psychiatry. 2022;13:941973. doi: https://doi.org/10.3389/fpsyt.....
6.
Nieto RR, Carrasco A, Corral S, Castillo R, Gaspar PA, Bustamante ML, Silva H. BDNF as a biomarker of cognition schizophrenia/psychosis: An updated review. Front Psychiatry. 2021;12:662407. doi: https://doi.org/10.3389/fpsyt.....
7.
Bothwell M. Recent advances in understanding context-dependent mechanisms controlling neurotrophin signalling and function. F1000 Faculty Rev-1658. 2019;8. doi: https://doi.org/10.12688/f1000....
8.
Deyama S, Bang E, Kato T, Li XY, Duman RS. Neurotrophic and antidepressant actions of brain-derived neurotrophic factor require vascular endothelial growth factor. Biological Psychiatry. 2019;86(2):143–152. doi: https://doi.org/10.1016/j.biop....
9.
Pijet B, Stefaniuk M, Kaczmarek L. MMP-9 contributes to dendritic spine remodeling following traumatic brain injury. Neural Plasticity. 2019;2019:3259295. doi: https://doi.org/10.1155/2019/3....
10.
Figiel I, Kruk PK, Zaręba-Kozioł M, Rybak P, Bijata M, Wlodarczyk J, Dzwonek J. MMP-9 signaling pathways that engage rho GTPases in brain plasticity. Cells. 2021;10(1):166. doi: https://doi.org/10.3390/cells1....
11.
Szepesi Z, Hostel E, Ruszczycky B, et al. Synaptically released matrix metalloproteinase activity in control of structural plasticity and the cell surface distribution of GluA1-AMPA receptors. PLoS One. 2014;9(5):e98274. doi: https://doi.org/10.1371/journa....
12.
Lepeta K, Kaczmarek L. Matrix metalloproteinase-9 as a novel player in synaptic plasticity and schizophrenia. Schizoph Bull. 2015;41(5):1003–1009. doi: https://doi.org/10.1093/schbul....
13.
Niitsu T, Ishima T, Yoshida T, Hashimoto T, Matsuzawa D, et al. A positive correlation between serum levels of mature brain-derived neurotrophic factor and negative symptoms in schizophrenia. Psychiatry Res. 2014;215(2):268–273. doi: https://doi.org/10.1016/j.psyc....
14.
Luo L, Li C, Deng Y, Wang Y, Meng P, Wang Q. High-intensity interval training on neuroplasticity, balance between brain-derived neurotrophic factor and precursor brain-derived neurotrophic factor in poststroke depression rats. J Stroke Cerebrovascular Dis. 2019;28(3):672–682. doi: https://doi.org/10.1016/j.stro....
15.
Li W, Kou C, Yu Y, et al. Association of catechol-o-methytransferase gene polymorphisms with schizophrenia and negative symptoms in a Chinese population. Am J Med Genet B Neuropsychiatr Genet. 2012;0(4):370–375. doi: https://doi.org/10.1002/ajmg.b....
16.
Kaczmarek L, Łapińska-Dzwonek J, Szymczak S. Matrix metalloproteinases in the adult brain physiology: A link between c-Fos, AP-1, and remodelling of neuronal connections? EMBO J. 2002;21(24):6643–6648. doi: https://doi.org/10.1093/emboj/....
17.
Xia QR, Zhang C, Liang J, Xu YY. The association of functional polymorphism of matrix metalloproteinase-9 gene (rs3918242) with schizophrenia: A meta-analysis. Int J Psychiatry Clin Practice. 2019;23(3):207–214. doi: https://doi.org/10.1080/136515....
18.
Lepeta K, Purzycka K, Pachulska-Wieczorek K, et al. A normal genetic variation modulates synaptic MMP-9 protein levels and the severity of schizophrenia symptoms. EMBO Molecular Med. 2017;9(8):1100–1116. doi: https://doi.org/10.15252/emmm.....
19.
Michaelsen K, Zagrebelsky M, Berndt-Huch J, et al. Neurotrophin receptors TrkB.T1 and p 75NTR cooperate in modulating both functional and structural plasticity in mature hippocampal neurons. Eur J Neurosci. 2010;32(11):1854–1865. doi: https://doi.org/10.1111/j.1460....
20.
Cyran A, Piotrowski P, Samochowiec J, Grąźlewski T, Misiak B. Risk factors of deficyt and non-deficit schizophrenia: Results from a cross-sectional study. Revista de Psiquiatria y Salud Mental (English Edition). 2022;15(4):223–229. doi: https://doi.org/10.1016/j.rpsm....
21.
Faden J, Citrome L. Resistance is not futile: Treatment-refractory schizophrenia-overview, evaluation, and treatment. Exp Opinion Pharmacotherapy. 2019;20(1):11–24. doi: https://doi.org/10.10180/14656....
22.
Markiewicz R. The use of EEG Biofeedback/Neurofeedback in psychiatric rehabilitation. Psychiatria Pol. 2017;51(6):1095–1106. doi: https://doi.org/10.12740/PP/68....
23.
Markiewicz R, Kozioł M, Olajossy M, Masiak J. Can the neurotrophic factor BDNF be an indicator of effective rehabilitation influences in schizophrenia? Psychiatria Pol. 2018;52(5):819–834. doi: https://doi. org/10.12740/PP/OnlineFirst/76040.
24.
Gandara V, Pineda JA, Shu IW, Singh F. A systematic review of the potential use of neurofeedback in patients with schizophrenia. Schizophrenia Bulletin. 2020;1(1):sgaa005. doi: https://doi.org/10.1093/schbul....
25.
Popova NK, Naumenko VS. Neuronal and behavioural plasticity: The role of serotonin and BDNF systems tandem. Expert Opinion on Therapeutic Targets. 2019;23(3):227–239. doi: https://doi.org/10.1080/ 14728222.2019.1572747.
26.
Mennerick S, Zorumski C. Neural activity, and survival in the developing nervous system. Mol Neurobiol. 2000;22(1–3):41–54. doi: https://doi.org/10.1385/MN:22:....
27.
Lefebvre S, Pavlidou A, Walther S. What is the potential of neurostumulation in the treatment of motor symptoms in schizophrenia? Expert Rev Neurotherapeutics. 2020;20(7):697–706. doi: https://doi.or g/10.1080/14737175.2020.1775586.
28.
Markiewicz-Gospodarek A, Markiewicz R, Dobrowolska B, Maciejewski R, Łoza B. Relationship of neuropeptide S with clinical and metabolic parameters of patients during rehabilitation therapy for schizophrenia. Brain Sci. 2022;12(6):768. doi: https://doi.org/10.3390/brains....
29.
Schulz KF, Altman DG, Moher D. CONSORT Group. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. Ann Intern Med. 2010;152(11):726–32. doi: https:// doi.org/10.7326/0003-4819-152-11-201006010-00232.
30.
Valle R. Schizophrenia in ICD-11: Comparsion of ICD-10 and DSM-5. Revista de Psiquiatria y Salud Mental (English Edition). 2020;13(2):95–104. doi: https://doi.org/10.1016/j.rpsm....
31.
Cohen J. Statistical power analysis for the behavioral sciences. 2nd ed. Hillsdale, New Jersey: Lawrence Erlabaum Associates, Publishers; 1988. ISBN 0-8058-083-5.
32.
Qu M, Wang J, Chen DC, Chen S, Xiu MH, Zhang XY. Sex-specific association between peripheral superoxide dismutase, BDNF, and cognitive impairment in drug-naïve first episode patients with schizophrenia. Free Radical Biol Med. 2020;160:887–893. doi: https:// doi.org/10.1016/j.freeradbiomed.2020.09.014.
33.
Yang F, Wang K, Du X, Deng H, Wu HE, et al. Sex difference in the association of body mass index and BDNF levels in Chinese patients with chronic schizophrenia. Psychopharmacol. 2019;236(2):753–762. doi: https://doi.org/10.1007/s00213....
34.
Yang Y, Zhang Y, Wang J, Ning X, et al. Sex differences in the association of HOMA-IR index and BDNF in Han Chinese patients with chronic schizophrenia. Front Psychiatry. 2021;12:656230. doi: https://doi. org/10.3389/fpsyt.2021.656230.
35.
Ali FT, Abd El-Azeem EM, Hamed MA, Ali MAM, Abd Al-Kader NM, Hassan EA. Redox dysregulation, immuno-inflammatory alterations, and genetic variants of BDNF and MMP-9 in schizophrenia: Pathophysiological and phenotypic implications. Schizophr Res. 2017;188:98–109. doi: https://doi.org/10.1016/j.schr....
36.
Collazos J, Asensi V, Martin G, Montes AH, Suárez-Zarracina T, Valle- Garay E. The effect of gender and genetic polymorphisms on matrix metalloprotease (MMP) and tissue inhibitor (TIMP) plasma levels in different infectious and non-infectious conditions. Clin Exp Immunol. 2015;182(2):213–219. doi: https://doi.org/10.1111/cei.12....
37.
Wei SM, Berman KF. Ovarian hormones, genes, and the brain: the case of estradiol and the brain-derived neurotrophic factor (BDNF) gene. Neuropsychopharmacology. 2019;44:223–224. doi: https://doi. org/10.1038/s41386-018-022305.
38.
Jaillon S, Berthenet K, Garlanda C. Sexual dimorphism in innate immunity. Clin Rev Allergy Immunol. 2019;56:308–321. doi: https:// doi.org/10.1007/s12016-017-8648-x.
39.
Takeuchi H, MacKenzie NE, Samaroo D, Agid O, Remington G, Leucht S. Antipsychotic dose in acute schizophrenia: A meta-analysis. Schizophrenia Bulletin. 2020;46(6):1439–1458. doi: https://doi. org/10.1093/schbul/sbaa063.
40.
Lim K, Peh OH, Yang Z, Rekhi G, Rapisarda A, See YM, et al. Large-scale evaluation of the Positive and Negative Syndrome Scale (PANSS) symptom architecture in schizophrenia. Asian J Psychiatry. 2021;62:102732. doi: https://doi.org/10.101 /j.ajp.2021.102732.
41.
Thompson M, Thompson L. The Neurofeedback Book, 2nd edition: An Introduction to Basic Concepts in Applied Psychophysiology. 2015,ISBN 10: 069292101X ISBN 13: 9780692921012, Association for Applied Psychophysiology and Biofeedback.
42.
Wykes T, Reeder C, Williams C, Corner J, Rice C, Everitt B. Are the effects of cognitive remediation therapy (CRT) durable? Results from an exploratory trial in schizophrenia. Schizophrenia Res. 2003;61:163–174. doi: https://doi.org/10.1016/S0920-....
43.
Markiewicz R, Dobrowolska B. Cognitive and social rehabilitation in schizophrenia – from neurophysiology to neuromodulation. Pilot Study. Int J Environ Res Public Health. 2020;17(11):4034. doi: https:// doi.org/10.3390/ijerph17114034.
44.
Markiewicz R, Dobrowolska B. Reinforcement of self-regulated brain activity in schizophrenia patients undergoing rehabilitation BioMed Res Int. 2021;Article ID 8030485, 1–9. doi: https://doi. org/10.1155/2021/8030485.
45.
Braithwaite J, Watson D, Jones R, Rowe M. A guide for analysing electrodermal activity (EDA) & and skin conductance responses (SCRs) for psychological experiments. Technical Report, 2nd version: Selective Attention & Awareness Laboratory (SAAL). Behavioural Brain Sciences Centre, University of Birmingham, UK, 2015.
46.
Lu EY, Cheng ASK, Tsang HWH, Chen J, Leung S, Yip A, et al. Psychoeducation, motivational interviewing, cognitive remediation training, and/or social skills training in combination for psychosocial functioning of patients with schizophrenia spectrum disorders: A systematic review and meta-analysis of randomized controlled trials. Front Psychiatry. 2022;13:899840. doi: https://doi.org/10.3389/ fpsyt.2022.899840.
47.
Markiewicz R, Markiewicz-Gospodarek A, Dobrowolska B, Łoza B. Improving clinical, cognitive, and psychosocial dysfunctions in patients with schizophrenia: A neurofeedback randomized control trial. Neural Plast. 2021;2021:4488664. doi: https://doi.org/10.1155/2021/4....
48.
Romash IR, Vynnyk MI. Peculiarities of brain-derived neurotrophic factor and matrix metalloproteinase-9 expression dynamics in patients with paranoid schizophrenia depending on the duration of the disease. Wiad Lek. 2021;74:2728–2732. PMID: 35023483.
49.
Arabska J, Margulska A, Strzelecki D, Wysokiński A. Does metabolic status affect serum levels of BDNF and MMP-9 in patients with schizophrenia? Nord J Psychiatry. 2019;73(8):515–521. doi: https:// doi.org/10.1080/08039488.2019.1658126.
50.
Vandooren J, Van den Steen PE, Opdenakker G. Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP- 9): The next decade. Critical Reviews in Biochemistry and Molecular Biology. 2013;48(3):222–272. doi: https://doi.org/10.3109/104092.... 2013.770819.
51.
Adams RA, Bush D, Zheng F, Meyer SS, Kaplan R, et al. Impaired theta phase coupling underlies frontotemporal dysconnectivity in schizophrenia. Brain. 2020;143(4):1261–1277. doi: https://doi. org/10.1093/brain/awaa035.
52.
Donkers FC, Schwikert SR, Evans AM, Cleary KM, Perkins DO, Belger A. Impaired neural synchrony in the theta frequency range in adolescents at familial risk for schizophrenia. Front Psychiatry. 2011;2:51. doi: https://doi.org/10.3389/fpsyt.....
53.
Lee HS, Kim JS. Implication of electrophysiological biomarkers in psychosis: Focusing on diagnosis and treatment response. J Pers Med. 2022;12(1):31. doi: https://doi.org/10.3390/jpm120....
54.
Atagun MI, Drukker M, Hall MH, Altun IK, Tatli SZ, Guloksuz S, van Os J, van Amelsvoort T. Meta-analysis of auditory P50 sensory gating in schizophrenia and bipolar disorder. Psych Res Neuroimaging. 2020;300:111078. doi: https://doi.org/10.1016/j. pscychresns.2020.11.111078.
55.
Javitt DC, Freedman R. Sensory processing dysfunction in the personal experience and neuronal machinery of schizophrenia. Am J Psychiatry.2015;172(1):17–31. doi: https://doi.org/10.1176/appi.a....
56.
Pachalska M, Góral-Pólrola J, Mueller A, Kropotov JD. Neuropsychology and the neurophysiology of perceptual microgenesis. Acta Neuropsychol. 2017;15(4):365–389. doi: 10.5604/01.3001.0010.7243.
57.
Kropotov JD. Functional neuromarkers for neuropsychology. Acta Neuropsychol. 2018;16(1):1–7. doi: 10.5604/01.3001.0011.6504.
58.
Loriette C, Ziane C, Ben Hamed S. Neurofeedback for cognitive enhancement and intervention and brain plasticity. Revue Neurologique. 2021;177(9):1133–1144.
59.
Luckos M, Cielebak K, Kaminski P. EEG neurofeedback in the treatment of cognitive dysfunctions after the infection of SARS-CoV-2 and long COVID-19. Acta Neuropsychol. 2021;19(3):361–372. doi: 10.5604/01.3001.0015.2464.
60.
Pazooki K, Leibetseder M, Renner W, Gougleris G, Kapsali E. Neurofeedback Treatment of Negative Symptoms in Schizophrenia: Two Case Reports. Appl Psychophysiol Biofeedback. 2019;44(1):31–39. doi: 10.1007/s10484-018-9417-1.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.