Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
REVIEW PAPER
Problems of Clostridium difficile infection (CDI) in Polish healthcare units
1
Chief Sanitary Inspectorate, Warsaw, Poland
2
Medical University of Lodz, Project InterDoktorMen, Poland
3
Military Institute of Hygiene and Epidemiology (WIHE), Laboratory of Epidemiology, Warsaw, Poland
4
Poznan University of Technology, Institute of Environmental Engineering and Building Installations, Poznan, Poland
Corresponding author
Zofia Kiersnowska
Chief Sanitary Inspectorate, Project InterDoktorMen, Medical University, Lodz, Poland
Chief Sanitary Inspectorate, Project InterDoktorMen, Medical University, Lodz, Poland
Ann Agric Environ Med. 2021;28(2):224-230
KEYWORDS
patient safetyrisk managementhospital-acquired infectionsHealthcare Associated InfectionClostridium difficile infections
TOPICS
ABSTRACT
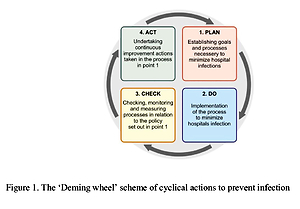
The issue of patient safety during the provision of health services poses a key challenge in health policy. The number of hospital-acquired infections (also known as HAI – Healthcare Associated Infection) determines the level of quality of health services provided in a given health facility. Effective management reinforced by the awareness of a team of medical professionals allows not only reduction in the hospital’s finances, but also the frequency of adverse events, which undoubtedly include hospital-acquired infections. Good cooperation between departments and a Hospital Infection Control Committee is one of the key aspects that translates to the rapid identification of new epidemic outbreaks. Infections caused by strains of Clostridium difficile (CDI, Clostridium difficile infection) are one of the main factors responsible for the prolonged hospitalization of patients. In the United States, Clostridium difficile causes almost half a million infections annually, and its treatment costs are estimated at nearly $ 4.8 billion per year. In Poland, the number of CDI cases in 2018 was 11.592 (for comparison, in 2013 the number of infections caused by this bacterium was 4.728). Hospital environment, inappropriate antibiotic therapy and development of multi-drug resistant strains increase the risk of infections. In order to improve the safety of hospitalized patients, infection risk management should be a systemic, formalized activity integrated with the overall process of managing a health facility. It is necessary that central units have interest in creating effective tools to enable successful epidemiological supervision and the implementation of strategic assumptions of health policy in this area.
REFERENCES (42)
1.
Dubberke ER, Olsen M.A. Burden of Clostridium difficile on the Healthcare System. Clin Infect Dis. 2012; 55(2): 88–92. doi: 10.1093/cid/cis335.
2.
Magill SS, Edwards JR, Bamberg W, Zintars G, at al. Multistate Point-Prevalence Survey of Health Care–Associated Infections. N Engl J Med. 2014; 370: 1198–1208. doi: 10.1056/NEJMoa1306801.
3.
NIZP-PZH Reports on cases of infectious diseases and poisonings in Poland http://wwwold.pzh.gov.pl/oldpa....
4.
Carroll R. Risk Management Handbook for Health Care Organizations. Hoboken. Wiley & Sons 2009; 78: 113.
5.
Center for Disease Control and Prevention. Healthcare-associated Infections (HAI). HAI Data:1.2018.https://www.cdc.gov/hai/data/i....
6.
Centers for Disease Control and Prevention. Antibiotic resistance threats in the United States. 2013: 11–21.
7.
Marczak M, et al. Risk Control in Health Care System, Methodology and Chosen Examples, Edited by Marczak M., Technical University of Lodz. 2008; 7.
8.
Ozorowski T. Zarządzanie ryzykiem zakażeń związanych z opieką zdrowotną. Forum zakażeń. 2018; 9(4): 203–207. doi: 10.15374/FZ2018032.
9.
Tworek P. Risk Management System as a Source of Knowledge About Hazards in Public Organizations in Poland. Studia Ekonomiczne. 2014; 199: 314–325.
10.
Turner NA, Smith BA, Lewis SS. Novel and emerging sources of Clostridioides difficile infection. PLoS Pathog. 2019; 15(12): e1008125. doi:10.1371/journal.ppat.1008125.
11.
Zdrowie i ochrona zdrowia w 2017 r. Analizy statystyczne. 100 lat GUS, Warszawa, Kraków 2018. ISSN 2084-0470.
13.
Michalski T. Ryzyko w działalności człowieka. Podstawy ubezpieczeń – Mechanizmy i funkcje, Wyd. Poltext, Warszawa. 2001; 1: 20–25.
14.
Coia J. C. difficile infection – Can we do better? Clin Microbiol Infect. 2018; 24(5): 450–451 doi: 10.1016/j.cmi.2017.12.009.
15.
Hamrol A, Mantura W. Zarządzanie jakością. Teoria i praktyka, PWN Warszawa. 2002; 200–210. https://www.nik.gov.pl/.
16.
Act on combating infections and infectious diseases in humans (Dz. U. z 2018 r. pos. 151, 1669); p.12.
17.
Regulation on the qualifications of members of the hospital infection control team (Dz. U. 2014 pos. 746); p.1.
18.
Paszko K. The role of the epidemiological nurse in the Infection Control Team. Forum Zakażeń 2011; 2(2): 57–60.
20.
Leitch M. ISO 31000:2009—The New International Standard on Risk Management. Risk Analysis, vol. 30, no. 6, 2010:887–892. doi: 10.1111/j.1539-6924.2010.01397.x
21.
Damani N. Risk management. IFIC Basic Concepts of Infection Control, 3rd edition, 2016.
22.
Badurek S, Muszytowski M, Stróżecki P, Manitius J. Clostridium difficile-associated disease in patients with chronic kidney disease. Forum Nefrol. 2016; 9(3): 141–148.
23.
He M, Miyajima F, Roberts P, et al. Emergence and global spread of epidemic healthcare-associated Clostridium difficile. Nat Genet. 2013; 45(1): 109–113. doi: 10.1038/ng.2478.
24.
Gateau C, Couturier J, Coia J, Barbut F. How to: diagnose infection caused by Clostridium difficile. Clin Microbiol Infect. 2018; 24(5): 463–468. doi: 10.1016/j.cmi.2017.12.005.
25.
Lemiech-Mirowska E, Kiersnowska Z, Ginter-Kramarczyk D, Kruszelnicka I. Toksynotwórcze szczepy Clostridium difficile-diagnostyka. Badania i rozwój młodych naukowców w Polsce 2019;3: 28–33.
26.
Pépin J, Routhier S, Gagnon S, Brazeau I. Management and outcomes of a first recurrence of Clostridium difficile-associated disease in Quebec, Canada. Clin Infect Dis. 2006; 42(6): 758–764. doi: 10.1086/501126.
27.
Martirosian G, Hryniewicz W, Ozorowski T, Pawlik K, Deptuła A. Zakażenia Clostridioides (Clostridium) difficile: epidemiologia, diagnostyka, terapia, profilaktyka 2018. Narodowy Program Ochrony Antybiotyków na lata 2016–2020.
28.
Marra AR, Perencevich EN, Nelson RE, et al. Incidence and Outcomes Associated With Clostridium difficile Infections: A Systematic Review and Meta-analysis. JAMA Netw Open. 2020; 3(1): e1917597. doi: 10.1001/jamanetworkopen.2019.17597.
29.
Marsh JW, Arora R, Schlackman JL, Shutt KA, Curry SR, Harrison LH. Association of relapse of Clostridium difficile disease with BI/NAP1/027. J Clin Microbiol. 2012; 50(12): 4078–82. doi: 10.1128/JCM.02291-12.
30.
Regulation the Minister of Health regarding the list of alert factors, hospital infection registers and alert factors as well as reports on the current epidemiological situation of the hospital (Dz. U. 2011 nr 294 poz. 1741).
31.
Jośko-Ochojska J, Spandel L. Clostridium difficile infections as a public health problem. Probl Hig Epidemiol. 2014; 95(3): 568–573.
32.
Ziółkowski G, Ziółkowska B, Ochocka B. Infekcje Clostridium difficile, niedoceniane zakażenia w aspekcie klinicznym i epidemiologicznym. Polskie Stowarzyszenie Pielęgniarek Epidemiologicznych, Katowice 2012.
33.
Albrecht P, Pituch H. Clostridium difficile — a growing diagnostic and therapeutic problem. Onkologia w Praktyce Klinicznej 2013; 9(1): 22–31.
34.
Zhang S, Palazuelos-Munoz S, Balsells EM, Nair H, Chit A, Kyaw MH. Cost of hospital management of Clostridium difficile infection in United States-a meta-analysis and modelling study. BMC Infect Dis. 2016; 16(1): 447. doi: 10.1186/s12879-016-1786-6.
35.
Czepiel J, Kędzierska J, Biesiada G, Birczyńska M, Perucki W, Nowak P, Garlicki A. Epidemiology of Clostridium difficile infection: results of a hospital-based study in Krakow, Poland. Epidemiol Infect. 2015; 143(15): 3235–43. doi: 10.1017/S0950268815000552.
36.
Desai K, Gupta SB, Dubberke ER, Prabhu VS, Browne C, Mast TC. Epidemiological and economic burden of Clostridium difficile in the United States: estimates from a modeling approach. BMC Infect Dis. 2016; 18(16): 303. doi: 10.1186/s12879-016-1610-3.
37.
Le Monnier A, Duburcq A, Zahar JR, Corvec S, Guillard T, Cattoir V, et al. Hospital cost of Clostridium difficile infection including the contribution of recurrences in French acute-care hospitals. J Hosp Infect. 2015; 91(2): 117–22. doi: 10.1016/j.jhin.2015.06.017.
38.
Zimlichman E1, Henderson D, Tamir O, Franz C, Song P, Yamin CK. Health care-associated infections: a meta-analysis of costs and financial impact on the US health care system. JAMA Intern Med. 2013; 173(22): 2039–46. doi: 10.1001/jamainternmed.2013.9763.
39.
Dubberke ER, Schaefer E, Reske KA, Zilberberg M, Hollenbeak CS, Olsen MA. Attributable inpatient costs of recurrent Clostridium difficile infections. Infect Control Hosp Epidemiol. 2014; 35(11): 1400–7. doi: 10.1086/678428.
40.
Jachowicz E, Pobiega M, Różańska A, Wójkowska-Mach J. Growing consumption of antibiotics and epidemiology of Clostridioides difficile infections in Poland: A need to develop new solutions. 2019; 9: 1–8. doi: 10.1556/030.66.2019.024.
41.
Albrecht P, Pituch H. Clostridium difficile — narastający problem diagnostyczny i terapeutyczny. Clostridium difficile — a growing diagnostic and therapeutic problem. Gastroenterol Klin. 2013; 5(1): 40–51.
42.
Ooijevaar RE, van Beurden YH, Terveer EM, et al. Update of treatment algorithms for Clostridium difficile infection. Clin Microbiol Infect. 2018; 24(5): 452–462. doi: 10.1016/j.cmi.2017.12.022.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.