Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
BRIEF COMMUNICATION
Improved Blastocystis spp. detection method using swabs with Amies transport medium and charcoal
1
Faculty of Medical Science. Collegium Medicum, Cardinal Stefan Wyszyński University in Warsaw, Poland
2
Polish Parasitological Society, Warsaw, Poland
3
Department of General Biology and Parasitology, Medical University of Warsaw, Poland
Corresponding author
Rusłan Sałamatin
Department of Parasitology. Medical University of Warsaw, Chałubińskiego 5, 02-004, Warsaw, Poland
Department of Parasitology. Medical University of Warsaw, Chałubińskiego 5, 02-004, Warsaw, Poland
Ann Agric Environ Med. 2022;29(2):303-305
KEYWORDS
TOPICS
ABSTRACT
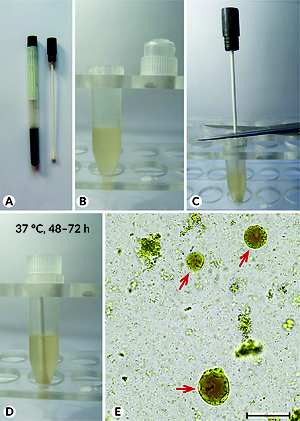
Blastocystis is one of the most frequently detected protozoa in the human large intestine. One of the most effective and cheap methods for detecting Blastocystis in faeces is culture on a special medium in anaerobic conditions. Sampling faeces using traditional containers and their transport to the laboratory has certain limitations: a sample taken in this way should reach the laboratory relatively quickly, moreover, some patients are uncomfortable during sampling and protection of material in this way. We propose utilizing a swab for sampling and transportation of the faeces samples to be examined for Blastocystis instead of using traditional containers. We believe this is an excellent method allowing the material to be transported over longer distances without additional, and sometimes expensive, safety measures, and at the same time permitting the possibility of obtaining living cells after a relatively long period of storage.
REFERENCES (8)
1.
Andersen LO, Stensvold CR. Blastocystis in health and disease: are we moving from a clinical to a public health perspective? J Clin Microbiol. 2016; 54(3): 524–528. doi: 10.1128/JCM.02520-15.
2.
Rudzińska M, Kowalewska B, Szostakowska B, Grzybek M, Sikorska K, Świątalska A. First report on the occurrence and subtypes of Blastocystis in pigs in Poland using sequence-tagged-site PCR and barcode region sequencing. Pathogens. 2020; 9(7): 595. doi: 10.3390/pathogens9070595.
3.
Can H, Köseoğlu AE, Erkunt Alak S, et al. Molecular prevalence and subtyping of Blastocystis sp. isolates in stray cats of İzmir, Turkey: first report of “ST4 allele 42” in cats. Pol J Vet Sci. 2021; 24(2): 217–223. doi: 10.24425/pjvs.2021.137656.
4.
Skotarczak B. Genetic diversity and pathogenicity of Blastocystis. Ann Agric Environ Med. 2018; 25(3): 411–416. doi: 10.26444/aaem/81315.
5.
Stensvold C. Laboratory diagnosis of Blastocystis spp. Trop Parasitol. 2015; 5(1): 3–5. doi: 10.4103/2229-5070.149885.
6.
Vaisusuk K, Saijuntha W, Sedlak S, et al. Blastocystis subtypes detected in long-tailed macaques in Thailand – further evidence of cryptic host specificity. Acta Trop. 2018; 184: 78–82. doi:10.1016/j.actatropica.2017.09.002.
7.
Amies CR. A modified formula for the preparation of Stuart’s transport medium. Can J Public Health. 1967; 58(7): 296–300.
8.
Jones WR. The experimental infection of rats with Entamoeba histolytica; with a method for evaluating the anti-amoebic properties of new compounds. Ann Trop Med Parasitol. 1946; 40(2): 130–140. doi: 10.1080/00034983.1946.11685270.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.