Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
CASE REPORT
Challenges in Acanthamoeba keratitis – treatment efficacy of a sight-threatening disease in a severe case without identified risk factors requiring diagnostic verification
1
First Department of Ophthalmology, Pomeranian Medical University, Szczecin, Poland
2
Department of Medical Biology,
The Medical University of Warsaw, Poland
3
SPKSO Ophthalmic Hospital, Warsaw, Poland
Corresponding author
Lidia Chomicz
Department of Medical Biology, The Medical University of Warsaw, Litewska 14/16, 00-575, Warszawa, Poland
Department of Medical Biology, The Medical University of Warsaw, Litewska 14/16, 00-575, Warszawa, Poland
Ann Agric Environ Med. 2021;28(4):719-723
KEYWORDS
Acanthamoeba keratitisdiagnostic techniques and proceduresconfocal microscopylight microscopyaxenic culturecombination drug therapy
TOPICS
- Biological agents posing occupational risk in agriculture, forestry, food industry and wood industry and diseases caused by these agents (zoonoses, allergic and immunotoxic diseases)
- Prevention of occupational diseases in agriculture, forestry, food industry and wood industry
- State of the health of rural communities depending on various factors: social factors, accessibility of medical care, etc.
ABSTRACT
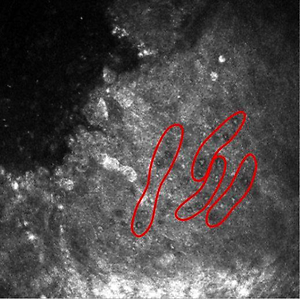
Acanthamoeba keratitis (AK), the vision-threatening disease caused by the amphizoic, potentially parasitic amoebae is growing threat for public health in Poland and worldwide. The report presents the case of 70-year-old man with severe keratitis admitted to an Ophthalmology Clinic. Before admission, the patient had been treated for 6 months with antibacterial and antifungal drugs in other units, without improvement in the eye condition. The use of in vivo confocal microscopy and in vitro cultivation allowed diagnosis to be verified and AK successfully treated. Awareness of the threat to public health caused by Acanthamoeba spp is still insufficient. If there is failure in response to first line therapy, AK should be taken into account,despite the lack of identified risk factors. In vitro monitoring of amoebic strain can be helpful for prognosis of the course of the corneal disease. Improvement in duration from first symptoms until proper diagnosis is decisive for better treatment efficacy.
REFERENCES (24)
1.
Marciano-Cabral F, Cabral G. Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev. 2003; 16(2): 273–307. https://doi.org/10.1128/cmr.16....
2.
Schuster FL, Visvesvara GS. Free-living amoebae as opportunistic and non-opportunistic pathogens of humans and animals. Int J Parasitol. 2004; 34: 1001–1027.
3.
McKelvie J, Alshiakhi M, Ziaei M, et al. The rising tide of Acanthamoeba keratitis in Auckland, New Zealand: a7-year review of presentation, diagnosis and outcomes (2009–2016). Clin Exp Ophthalmol. 2018; 46: 60 0 – 607. ht t ps://doi.org /10.1111/ceo.13166.
4.
Hajialilo E, Niyyati M, Solaymani M, et al. Pathogenic free-living amoebae isolated from contact lenses of keratitis patients. Iran J Parasitol. 2015; 10(4): 541–546.
5.
Walochnik J, Scheikl U, Haller-Schober EM. Twenty years of Acanthamoeba diagnostics in Austria. J Eukaryot Microbiol. 2015; 62: 3 –11. ht t ps://doi.org /10.1111/jeu.12149.
6.
Huang,FC, Shih MH, Chang KF, et al. Characterizing clinical isolates of Acanthamoeba castellanii with high resistance to polyhexamethylene biguanide in Taiwan. J Microbiol Immunol Infect. 2017; 50: 570–577. doi: S1684-1182(15)00908-1 [pii].1155/2015/231285.
7.
Chomicz L, Conn DB, Padzik M, et al. Emerging threats for human health in Poland: pathogenic isolates from drug resistant Acanthamoeba keratitis monitored in terms of their in vitro dynamics and temperature adaptability. BioMed Res Int. 2015, ID231285. https://doi.org/10.
8.
Król-Turmińska K, Olender A. Human infections caused by free-living amoebae. Ann Agric Environ Med. 2017; 24(2): 254–260. doi: 10.5604/12321966.1233568.
9.
Khan NA. Acanthamoeba: biology and pathogenesis. 2nd ed. Caister Academic Press; 2015.
10.
Chomicz L, Szaflik JP, Padzik M, Izdebska J. Acanthamoeba keratitis: the emerging vision threatening corneal disease. In: Advances in Common Eye Infections. (Ed. S. Rumelt). INTECH. 2016, Chapter 4, p. 99–120.
11.
Somani SN, Ronquillo Y, Moshirfar M. Acanthamoeba Keratitis. In Stat. Pearls. Publishing LLC: Treasure Island, FL, USA, 2020.
12.
Shimmura-Tomita M, Takano H, Kinoshita N, et al. Risk factors and clinical signs of severe Acanthamoeba keratitis. Clin Ophthalmol. 2018; 12: 2567–2573.
13.
Lorenzo-Morales J, Khan NA, Walochnik J. An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite. 2015; 22(10): 1–20. https://doi.org/10.1051/parasi....
14.
Garg P, Kalra P, Joseph J. Non-contact lens related Acanthamoeba keratitis. Indian J Ophthalmol. 2017; 65(11): 1079–1086. https://doi.org/10.4103/ijo.IJ..._ 826 _17.
15.
Szentmary N, Daas L, Shi L, et al. Acanthamoeba keratitis – Clinical signs, differential diagnosis and treatment. J Current Ophthalmol. 2019; 31: 16–23.
16.
Juárez MM, Tártara LI, Cid AG, et al. Acanthamoeba in the eye, can the parasite hide even more? Latest developments on the disease. Contact Lens & Anterior Eye. 2018; 41: 245–251. doi: 10.1016/j.clae.2017.12.017.
17.
Villani E, Baudouin C, Efron N, et al. In vivo confocal microscopy of the ocular surface: from bench to bedside. Curr. Eye. Res. 2014; 39: 213–231. doi: 10.3109/02713683.2013.842592.
18.
Padzik M, Szaflik JP, Baltaza W, et al. In vivo confocal microscopy and in vitro culture techniques as tools for evaluation of severe Acanthamoeba keratitis incidents. Ann Parasitol. 2017; 63(4): 341–346.
19.
Chomicz L, Padzik M, Szaflik JP, et al. Monitoring of in vitro dynamics of Acanthamoeba strains isolated from infected eyes as an useful tool in keratitis management. Exp Parasitol. 2014; 145: 73–77.
20.
Červa L, Novak K. Amoebic meningoencephalitis: sixteen fatalities. Science. 1968; 160: 92.
21.
Sepehr Feizi. Corneal Angiogenesis: Etiologies, Complications, and Management, In: Physiologic and Pathologic Angiogenesis – Signaling Mechanisms and Targeted Therapy, (Ed. Dan Simionescu and Agneta Simionescu), IntechOpen. 2017. Available at: https://www.intechopen.com/boo....
22.
Bock F, Konig Y, Kruse F, et al. Bevacizumab (Avastin) eye drops inhibit corneal neovascularization. Graefe’s Arch Clin Exp Ophthalmol. 2008; 246(2): 281–284. https://doi.org/10.1007/s00417....
23.
Koenig Y, Bock F, Horn F, et al. Short- and long-term safety profile and efficacy of topical bevacizumab (Avastin) eye drops against corneal neovascularization. Graefe’s Arch Clin Exp Ophthalmol. 2009; 247(10): 1375–1382. https://doi.org/10.1007/s00417....
24.
Robaei D, Carnt N, Minassian DC, et al. The impact of topical corticosteroid use before diagnosis on the outcome of Acanthamoeba keratitis. Ophthalmol. 2014; 121(7): 1383–1388. https://doi.org/10.1016/j.opht....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.