Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Intraoral microbiome components identified in Polish patients assessed in terms of threats to human health with infectious factors
1
Department of Orthodontics, Medical University, Warsaw, Poland
2
Department of General Biology and Parasitology, Medical University, Warsaw, Poland
3
Department of Oral Surgery, Medical University of Warsaw, Poland
4
Department of Ophthalmology, Independent Public Clinical Ophthalmology Hospital, Medical University, Warsaw, Poland
5
AGAMED Agnieszka Łukomska-Kłosok, Szczecin, Poland
6
Department of Invertebrate Zoology, Museum of Comparative Zoology, Harvard University, Cambridge, United States
7
One Health Center, Berry College, Mount Berry, Georgia, United States
8
Clinic of Cranio-Maxillo-Facial and Oral Surgery and Implantology, Medical University, Warsaw, Poland
9
Department of Tropical Parasitology, Faculty of Health Sciences, Medical University, Gdańsk, Poland
10
Department of Public Health, Medical University, Warsaw, Poland
11
Department of Medical Biology, Medical University, Warsaw, Poland
Corresponding author
Lidia Chomicz
Department of Medical Biology, Medical University of Warsaw, Litewska 14/16, 00-575, Warszawa, Poland
Department of Medical Biology, Medical University of Warsaw, Litewska 14/16, 00-575, Warszawa, Poland
KEYWORDS
intraoral microbiomeinfectious factorsparasitic oral protistsamphizoic amoebaeopportunistic and pathogenic bacteria and fungiincreasing threats to human health
TOPICS
- Biological agents posing occupational risk in agriculture, forestry, food industry and wood industry and diseases caused by these agents (zoonoses, allergic and immunotoxic diseases)
- Prevention of occupational diseases in agriculture, forestry, food industry and wood industry
- State of the health of rural communities depending on various factors: social factors, accessibility of medical care, etc.
ABSTRACT
Introduction and objective:
The human oral cavity, the main part of masticatory system, is a dynamic environment still requiring quality research. The aim of the study is assessment of the status of the oral cavity and composition of intraoral microbiome of Polish patients in terms of threats to human health with infectious factors.
Material and methods:
The study utilised the data of generally healthy persons: 30 young aged 16–26 years and 30 middle-aged patients, aged 42–52 years. Intraoral swabs were assessed microscopically and by in vitro culture methods to detect/ identify microbiota.
Results:
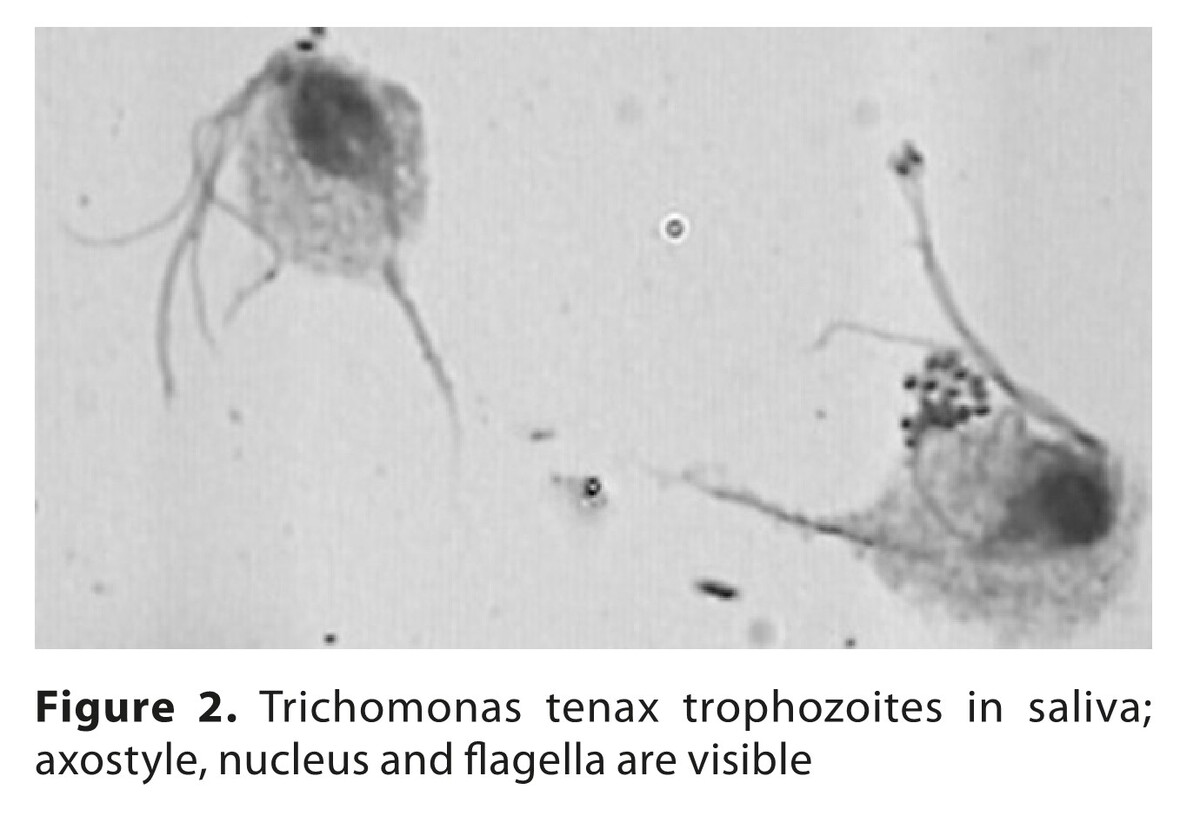
Different microorganisms occurr in the oral cavity, including non-resident species. Parasitic protozoans Trichomonas tenax and Entamoeba gingivalis, facultative parasitic Acanthamoeba strains, yeast-like fungi of Candida albicans group, opportunistic and pathogenic bacteria, including endosymbionts, were identified with various frequency in particular regions of the oral cavity. Higher prevalences of bacteria and fungi strains occurred in middle-aged patients.
Conclusions:
The relationship between microbiota of the human oral cavity remains a rare subject of research. This study has shown the ability of different microorganisms to coexist intraorally. These components may pose clinically important threat that should be taken into account as infectious factors. Recognition of microbiome components as potentially contagious, early identification/monitoring/assessment of concomitant species, preventive elimination of the infectious strains during the treatment should be taken into consideration. Further quality research on the intraoral microbiome species that may pose severe local/general clinical diseases are needed to reduce the risk to human health.
The human oral cavity, the main part of masticatory system, is a dynamic environment still requiring quality research. The aim of the study is assessment of the status of the oral cavity and composition of intraoral microbiome of Polish patients in terms of threats to human health with infectious factors.
Material and methods:
The study utilised the data of generally healthy persons: 30 young aged 16–26 years and 30 middle-aged patients, aged 42–52 years. Intraoral swabs were assessed microscopically and by in vitro culture methods to detect/ identify microbiota.
Results:
Different microorganisms occurr in the oral cavity, including non-resident species. Parasitic protozoans Trichomonas tenax and Entamoeba gingivalis, facultative parasitic Acanthamoeba strains, yeast-like fungi of Candida albicans group, opportunistic and pathogenic bacteria, including endosymbionts, were identified with various frequency in particular regions of the oral cavity. Higher prevalences of bacteria and fungi strains occurred in middle-aged patients.
Conclusions:
The relationship between microbiota of the human oral cavity remains a rare subject of research. This study has shown the ability of different microorganisms to coexist intraorally. These components may pose clinically important threat that should be taken into account as infectious factors. Recognition of microbiome components as potentially contagious, early identification/monitoring/assessment of concomitant species, preventive elimination of the infectious strains during the treatment should be taken into consideration. Further quality research on the intraoral microbiome species that may pose severe local/general clinical diseases are needed to reduce the risk to human health.
REFERENCES (30)
1.
Jenkinson HF, Lamont RJ. Oral microbial communities in sickness and in health. Trends Microbiol. 2005;13(12):589–595. doi:10.1016/j.tim.2005.09.006.
2.
Demling A, Heuer W, Elter C, at al. Analysis of supra- and subgingival long-term biofilm formation on orthodontic bands. Eur J Orthod. 2009;31:202–206.
3.
Zaura E, Keijser BJ, Huse SM, et al. Defining the healthy “core microbiome” of oral microbial communities. BMC Microbiol. 2009;9(1):259–270. doi:10.1186/1471-2180-9-259.
4.
Chomicz L, Perkowski K, Siemińska-Piekarczyk B, et al. Assessment of various components of oral cavity ontocenosis as potential factors for pre and post-surgery complications. II. Opportunistic fungi and protozoans in the oral cavity of orthodontic patients. Chir Czaszk Szczęk Twarz Ortop Szczęk. 2009;4:67–76.
5.
Ghannoum MA, Jurevic RJ, Mukherjee PK, et al. Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathogens. 2010;6(1), Article ID e1000713. doi:10.1371/journal.ppat.1000713.
6.
Wróblewska M, Strużycka I, Mierzwińska-Nastalska E. Significance of biofilms in dentistry. Przegl Epidemiol. 2015;69(4):739–744.
7.
Mahdi SS, Sibilio F, Amenta F. Dental hygiene habits and oral health status of seafarers. Int Marit Health. 2016;67(1):9–13. doi:10.5603/ IMH.2016.0003.
8.
Filoche S, Wong L, Sissons CH. Oral biofilms: emerging concepts in microbial ecology. J Dent Res. 2010;89:8–18. doi:10.1177/0022034509351812.
9.
Marsh PD. Dental plaque: biological significance of a biofilm community lifestyle. J Clin Periodontol. 2005;32:7–15.
10.
Thuy D, Devine D, Marsh P. Oral biofilms: molecular analysis, challenges, and future prospects in dental diagnostics. Clin Cosmet Investig Dent. 2013;5:11–19. doi:10.2147/CCIDE.S31005.
11.
Zawadzki PJ, Perkowski K, Starościak B, et al. Evaluation of selected oral cavity microbiota – risk factors of management complications in patients with masticatory system disorders. Ann Parasitol. 2016;62(1):71–76.
12.
Doern CD, Burnham CA. It’s not easy being green: the viridans group streptococci, with a focus on pediatric clinical manifestations. J Clin Microbiol. 2010;48(11):3829–3835.
13.
Przybyłowska D, Mierzwińska-Nastalska E, Swoboda-Kopeć E, et al. Potential respiratory pathogens colonization of the denture plaque of patients with chronic obstructive pulmonary disease. Gerodontology. 2016;33:322–327. doi:10.1111/ger.12156.
14.
Zawadzki PJ, Perkowski K, Padzik M, et al. Examination of oral microbiota diversity in adults and older adults as an approach to prevent spread of risk factors for human infections. BioMed Res Int. 2017; Article ID 8106491.7 pages. doi: 10.1155/2017/8106491.
15.
Bik EM, Long CD, Armitage GC, et al. Bacterial diversity in the oral cavity of 10 healthy individuals. ISME J. 2010;4:962–974. doi:10.1038/ismej.2010.30.
16.
Belda-Ferre P, Alcaraz LD, Cabrera-Rubio R, et al. The oral metagenome in health and disease. The ISME J. 2012;6:46–56 doi:10.1038/ismej.2011.85.
17.
Perkowski K, Baltaza W, Conn DB, et al. Examination of oral biofilm microbiota in patients using fixed orthodontic appliances in order to prevent risk factors for health complications. Ann Agric Environ Med. 2019;26(2):231–235. doi:10.26444/aaem/105797.
18.
Downes J, Mantzourani M, Beighton D, et al. Scardovia wiggsiae sp. nov., isolated from the human oral cavity and clinical material, and emended descriptions of the genus Scardovia and Scardovia inopinata. Int J Syst Evol Microbiol. 2011;61:25.
19.
Perkowski K, Chomicz L, Starościak B, et al. Occurrence of pathogenic bacteria in the oral cavity of the orthodontic patients requiring surgical treatment. Stomatol Współcz. 2012;19:8–13.
20.
Zawadzki PJ, Perkowski K, Starościak B, et al. Identification of infectious microbiota from oral cavity environment of various population group patients as a preventive approach to human health risk factors. Ann Agric Environ Med. 2016;23(4):675–678. doi:10.5604/12321966.1226847.
21.
Chomicz L, Szaflik J.P, Kuligowska A, et al. Concomitant Potentially Contagious Factors Detected in Poland and Regarding Acanthamoeba Strain etiological Agents of Keratitis in Humans. Microorganisms. 2024;12(12). https://doi.org/10.3390/microo....
22.
Trofa D, Gacser A, Nosanchuk JD. Candida parapsilosis, an emerging fungal pathogen. Clin Microbiol Rev. 2008;(4):606–625. doi:10.1128/CMR.00013-08.
23.
Chomicz L, Piekarczyk J, Starościak B, et al. Comparative studies on the occurrence of protozoans, bacteria and fungi in the oral cavity of patients with systemic disorders. Acta Parasitol. 2016;47:147–153C.
24.
Molmeret M, Horn M, Wagner M, et al Amoebae as training grounds for intracellular bacterial pathogens. Appl Environ Microbiol. 2005;71:20–28.
25.
Trabelsi H, Sellami A, Dendena F, et al. A. free-living amoebae (FLA): Morphological and molecular identification of Acanthamoeba in dental unit water. Parasite. 2010;17:67–70. doi:10.1051/parasite/2010171067.
26.
Lorenzo-Morales J, Khan NA, Walochnik J. An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite. 2015;22:1–20. https://doi.org/10.1051/parasi....
27.
Chomicz L, Szaflik JP, Padzik M, Izdebska J. Acanthamoeba keratitis: The emerging vision – threatening corneal disease. Advances in Common Eye Infections. Ed.: Shimon Rumelt. Published by INTECH 2016;2:99–120.
28.
Kowalski RP, Nayyar SV, Romanowski EG, et al. The prevalence of Bacteria, Fungi, Viruses, and Acanthamoeba From 3,004 Cases of Keratitis, Endophthalmitis, and Conjunctivitis. Eye Contact Lens 2020;46:5–268. [CrossRef].
29.
Lunsford RD, Melillo AA, Somerman MJ. Guest editorial for special oral microbes edition. Microbes Infect. 2015;17(7):471–472.
30.
Iovieno A, Ledee DR, Miller D, Alfonso EC, Detection of bacterial endosymbionts in clinical Acanthamoeba isolates. Ophthalmol. 2010;117:445–452.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.