Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
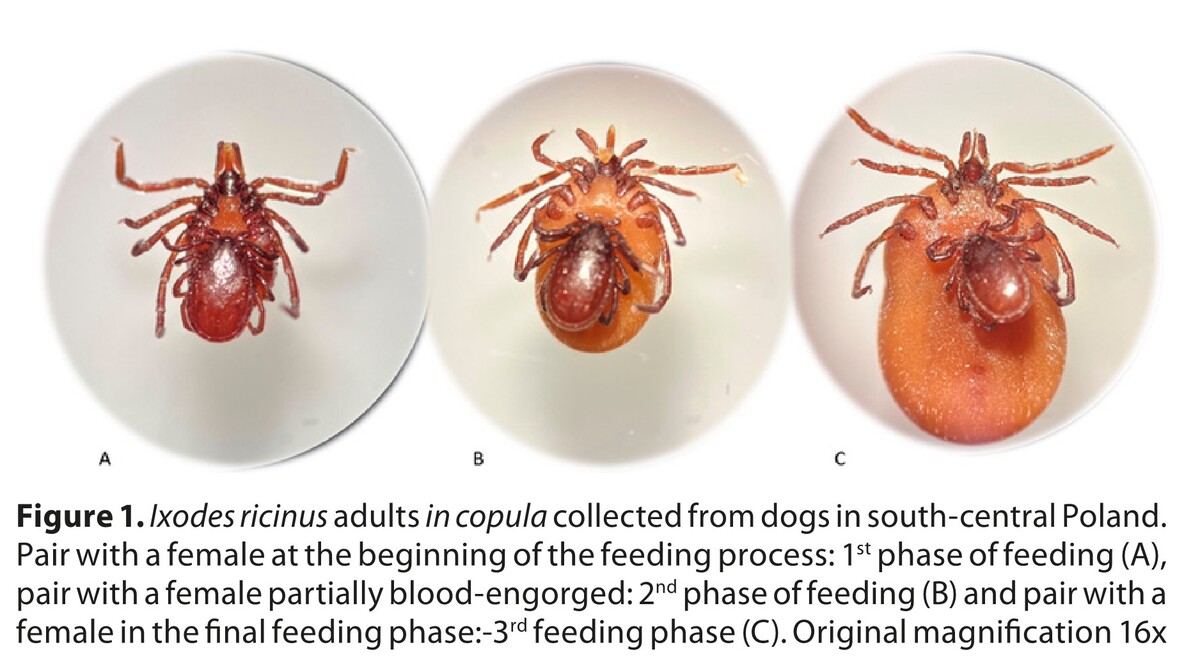
Copulation of Ixodes ricinus males and females on the host and its potential impact on pathogen transmission
1
Medical University, Lublin, Poland
2
Medical University of Silesia, Katowice, Poland
Ann Agric Environ Med. 2023;30(4):617-622
KEYWORDS
Borrelia burgdorferiAnaplasma phagocytophilumIxodes ricinuspathogen transmissiontick-borne pathogenstick copulation
TOPICS
ABSTRACT
Introduction and objective:
The common tick Ixodes ricinus is one of Europe’s most important vectors of tick-borne diseases. The increased risk of attacks by this tick suggests the need for identification of factors contributing to the transmission of tick-borne pathogens, and the routes of pathogen circulation in nature.
Material and methods:
Polymerase chain reaction was used to investigate the prevalence of four pathogens, i.e. Borrelia burgdorferi s.l. (Bb), Anaplasma phagocytophilum (Ap), Babesia spp. (Bs), and Bartonella spp. (Ba) in I. ricinus females and males mating on dogs in south-central Poland.
Results:
The study revealed the presence of three pathogens: Bb, Ap, and Bs in 9.4%, 5.4%, and 5.4% of all I. ricinus adults in copula, respectively. Co-infection with two pathogens was detected in one tick specimen. Borrelia burgdorferi spirochetes were isolated in two females and two males in copula, but the sexual transfer of the spirochetes between these specimens could not be clearly confirmed.
Conclusions:
By increasing the feeding dynamics in females, the copulation of I. ricinus males with females attached to the host’s skin may stimulate pathogen replication in tick tissues and migration from the gut to the salivary glands. Further investigations of the I. ricinus copulation on the host on female feeding and pathogen transmission may contribute to the elucidation of the eco-epidemiology of tick-borne diseases transmitted by this tick species.
The common tick Ixodes ricinus is one of Europe’s most important vectors of tick-borne diseases. The increased risk of attacks by this tick suggests the need for identification of factors contributing to the transmission of tick-borne pathogens, and the routes of pathogen circulation in nature.
Material and methods:
Polymerase chain reaction was used to investigate the prevalence of four pathogens, i.e. Borrelia burgdorferi s.l. (Bb), Anaplasma phagocytophilum (Ap), Babesia spp. (Bs), and Bartonella spp. (Ba) in I. ricinus females and males mating on dogs in south-central Poland.
Results:
The study revealed the presence of three pathogens: Bb, Ap, and Bs in 9.4%, 5.4%, and 5.4% of all I. ricinus adults in copula, respectively. Co-infection with two pathogens was detected in one tick specimen. Borrelia burgdorferi spirochetes were isolated in two females and two males in copula, but the sexual transfer of the spirochetes between these specimens could not be clearly confirmed.
Conclusions:
By increasing the feeding dynamics in females, the copulation of I. ricinus males with females attached to the host’s skin may stimulate pathogen replication in tick tissues and migration from the gut to the salivary glands. Further investigations of the I. ricinus copulation on the host on female feeding and pathogen transmission may contribute to the elucidation of the eco-epidemiology of tick-borne diseases transmitted by this tick species.
ACKNOWLEDGEMENTS
We are grateful to Maciej Rudek PhD for his support in tick
collection.
REFERENCES (97)
1.
ECDC. European Centre for Disease Prevention and Control and European Food Safety Authority. Ixodes ricinus – current known distribution: May 2020. Stockholm. 2020. https://www.ecdc.europa.eu/en/....
2.
Jongejan F, Uilenberg, G. The global importance of ticks. Parasitology. 2004;129(Suppl.):S3–S14. https://doi.org/10.1017/S00311....
3.
Wójcik-Fatla A, Szymańska J, Wdowiak L, Buczek A, Dutkiewicz J. Coincidence of three pathogens (Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum and Babesia microti) in Ixodes ricinus ticks in the Lublin macroregion. Ann Agric Environ Med. 2009;16(1):151–158.
4.
Pettersson JHO, Golovljova I, Vene S, et al. Prevalence of tick-borne encephalitis virus in Ixodes ricinus ticks in northern Europe with particular reference to Southern Sweden. Parasit Vectors. 2014;7:102. https://doi.org/10.1186/1756-3....
5.
Bertola M, Montarsi F, Obber F, Da Rold G, Carlin S, Toniolo F, Porcellato E, Falcaro C, Mondardini V, Ormelli S, Ravagnan S. Occurrence and identification of Ixodes ricinus borne pathogens in Northeastern Italy. Pathogens. 2021;10(9):1181. https://doi.org/10.3390/pathog....
6.
Markowicz M, Schötta AM, Höss D, et al. Infections with tickborne pathogens after tick bite, Austria, 2015–2018. Emerg Infect Dis. 2021;27(4):1048–1056. https://doi.org/10.3201/eid270....
7.
Olsthoorn F, Sprong H, Fonville M, et al. Occurrence of tick-borne pathogens in questing Ixodes ricinus ticks from Wester Ross, Northwest Scotland. Parasit Vectors. 2021;14:430. https://doi.org/10.1186/s13071....
8.
Kubiak K, Szymańska H, Dmitryjuk M, Dzika E. Abundance of Ixodes ricinus Ticks (Acari: Ixodidae) and the diversity of Borrelia species in Northeastern Poland. Int J Environ Res Public Health. 2022;19(12):7378. https://doi.org/10.3390/ijerph....
9.
Kunze U. The International Scientific Working Group on Tick-Borne Encephalitis (ISW TBE): review of 17 years of activity and commitment. Ticks Tick Borne Dis. 2016;7(3):399–404.
10.
van den Wijngaard CC, Hofhuis A, Simoes M, et al. Surveillance perspective on Lyme borreliosis across the European Union and European Economic Area. Eurosurveillance. 2017;22. https://doi.org/10.2807/1560-7....
11.
European Parliament. European Parliament resolution on Lyme disease (Borreliosis) (2018/2774(RSP)). 2018.
12.
Adaszek Ł, Winiarczyk S, Kutrzeba J, Puchalski A, Dębiak P. Przypadki boreliozy u psów na Lubelszczyźnie. Życie Wet. 2008b;83:311–313.
13.
Beugnet F, Marie JL. Emerging arthropod borne diseases of companion animals in Europe. Vet Parasitol. 2009;163:298–305.
14.
Bajer A, Beck A, Beck R, et al. Babesiosis in Southeastern, Central and Northeastern Europe: an emerging and re-emerging tick-borne disease of humans and animals. Microorganisms. 2022;10:945. https://doi.org/10.3390/microo....
15.
Bartosik K, Sitarz M, Szymańska J, Buczek A. Tick bites on humans in the agricultural and recreational areas in south-eastern Poland. Ann Agric Environ Med. 2011;18(1):151–157.
16.
Wilhelmsson P, Lindblom P, Fryland L, et al. Ixodes ricinus ticks removed from humans in Northern Europe: seasonal pattern of infestation, attachment sites and duration of feeding. Parasit Vectors. 2013;6:362. https://doi.org/10.1186/1756-3....
17.
Buczek W, Buczek AM, Bartosik K, Buczek A. Comparison of Skin Lesions Caused by Ixodes ricinus Ticks and Lipoptena cervi Deer Keds Infesting Humans in the Natural Environment. Int J Environ Res Public Health. 2020;17(9):3316. https://doi.org/10.3390/ijerph....
18.
Pawełczyk A, Bednarska M, Hamera A, et al. Long-term study of Borrelia and Babesia prevalence and co-infection in Ixodes ricinus and Dermacentor recticulatus ticks removed from humans in Poland, 2016–2019. Parasit Vectors. 2021;14(1):348. https://doi.org/10.1186/s13071....
19.
Hamsten C, Starkhammar M, Tran TA, et al. Identification of galactose-?-1,3-galactose in the gastrointestinal tract of the tick Ixodes ricinus; possible relationship with red meat allergy. Allergy. 2013;68(4):549–552. doi:10.1111/all.12128.
20.
Fischer J, Lupberger E, Hebsaker J, et al. Prevalence of type I sensitization to alpha-gal in forest service employees and hunters. Allergy. 2017;72(10):1540–1547. https://doi.org/10.1111/all.13....
21.
Rutkowski K, Sowa P, Mroczko B, et al. Sensitisation and allergic reactions to alpha-1,3-galactose in Podlasie, Poland, an area endemic for tick-borne infections. Infect Dis (Lond). 2022;54(8):572–579. https://doi.org/10.1080/237442....
22.
Banović P, Díaz-Sánchez AA, Galon C, et al. Humans infested with Ixodes ricinus are exposed to a diverse array of tick-borne pathogens in Serbia. Ticks Tick Borne Dis. 2021;12(2):101609. https://doi.org/10.1016/j.ttbd....
23.
Jumpertz M, Sevestre J, Luciani L, Houhamdi L, Fournier PE, Parola P. Bacterial agents detected in 418 ticks removed from humans during 2014–2021, France. Emerg Infect Dis. 2023;29(4):701–710. doi:10.3201/eid2904.221572.
24.
Földvári G, Márialigeti M, Solymosi N, et al. Hard ticks infesting dogs in Hungary and their infection with Babesia and Borrelia species. Parasitol Res. 2007;101:25–34. doi:10.1007/s00436-007-0608-6.
25.
Claerebout E, Losson B, Cochez C, et al. Ticks and associated pathogens collected from dogs and cats in Belgium. Parasit Vectors. 2013;6:183. https://doi.org/10.1186/1756-3....
26.
Król N, Obiegala A, Pfeffer M, et al. Detection of selected pathogens in ticks collected from cats and dogs in the Wrocław agglomeration, South-West Poland. Parasit Vectors. 2016;9:351. https://doi.org/10.1186/s13071....
27.
Abdullah S, Helps C, Tasker S, et al. Ticks infesting domestic dogs in the UK: a large-scale surveillance programme. Parasit Vectors. 2016;9:391. https://doi.org/10.1186/s13071....
28.
Geurden T, Becskei C, Six RH, et al. Detection of tick-borne pathogens in ticks from dogs and cats in different European countries. Ticks Tick Borne Dis. 2018;9(6):1431–1436. https://doi.org/10.1016/j.ttbd....
29.
Kocoń A, Nowak-Chmura M, Asman M. Assessment of the species composition of ticks attacking dogs and domestic cats in the Małopolska and Silesian provinces. Syst Appl Acarol. 2022;27(8):1509–1517. https://doi.org/10.11158/saa.2....
30.
Probst J, Springer A, Strube C. Year-round tick exposure of dogs and cats in Germany and Austria: results from a tick collection study. Parasit Vectors. 2023;16(1):70. https://doi.org/10.1186/s13071....
31.
Lejal E, Marsot M, Chalvet-Monfray K, et al. A three-years assessment of Ixodes ricinus-borne pathogens in a French peri-urban forest. Parasit Vectors. 2019;12(1):551. https://doi.org/10.1186/s13071....
32.
Kubiak K, Dziekońska-Rynko J, Szymańska H, Kubiak D, Dmitryjuk M, Dzika E. Questing Ixodes ricinus ticks (Acari, Ixodidae) as a vector of Borrelia burgdorferi sensu lato and Borrelia miyamotoi in an urban area of north-eastern Poland. Exp Appl Acarol. 2019;78(1):113–126. https://doi.org/10.1007/s10493....
33.
Grochowska A, Milewski R, Pancewicz S, et al. Comparison of tick-borne pathogen prevalence in Ixodes ricinus ticks collected in urban areas of Europe. Sci Rep. 2020;10(1):1–9. https://doi.org/10.1038/s41598....
34.
Hauck D, Springer A, Chitimia-Dobler L, Strube C. Two-year monitoring of tick abundance and influencing factors in an urban area (city of Hanover, Germany). Ticks Tick Borne Dis. 2020;11(5):101464. https://doi.org/10.1016/j.ttbd....
35.
Vogelgesang JR, Walter M, Kahl O, Rubel F, Brugger K. Long-term monitoring of the seasonal density of questing ixodid ticks in Vienna (Austria): Setup and first results. Exp Appl Acarol. 2020;81(3):409–420. https://doi.org/10.1007/s1049 3-020-00511-4.
36.
Hansford KM, Wheeler BW, Tschirren B, Medlock JM. Questing Ixodes ricinus ticks and Borrelia spp. in urban green space across Europe: A review. Zoonoses Public Health. 2022;69:153–166.
37.
Bartosik K, Buczek A. The impact of intensity of invasion of Ixodes ricinus and Dermacentor reticulatus on the course of the parasitic phase. Ann Agric Environ Med. 2012;19(4):651–655.
38.
Honzáková E, Olejniček J, Čern? V, Daniel M, Dusbábek F. Relationship between number of eggs deposited and body weight of engorged Ixodes ricinus female. Folia Parasitol (Praha). 1975;22:37–43.
39.
Bowessidjaou J, Brossard M, Aeschlimann A. Effects and duration of resistance acquired by rabbits on feeding and egg laying in Ixodes ricinus L. Cell Mol Life Sci. 1977;33(4):528–530.
40.
Schorderet S, Brossard M. Changes in immunity to Ixodes ricinus by rabbits infested at different levels. Med Vet Entomol. 1993;7(2):186–192. doi:10.1111/j.1365-2915.1993.tb00673.x.
41.
Buczek A, Barańska E, Buczek W, Bartosik K, Ciura D, Buczek AM. Preoviposition and oviposition of female Ixodes ricinus (L., 1758) (Ixodida: Ixodidae) at different levels of humidity under laboratory conditions. Ann. Parasitol. 2022;(68):s247.
42.
Bartosik K, Buczek A, Borzęcki A, Kulina D. Study of the non-parasitic stage in Ixodes ricinus after co-feeding with Dermacentor reticulatus in three infestations. Ann Agric Environ Med. 2017;24(1):90–95. doi: 10.5604/12321966.1234005.
43.
Alekseev AN, Chunikhin SP. The exchange of the tick-borne encephalitis virus between ixodid ticks feeding jointly on animals with a subthreshold level of viremia. Med Parazitol. 1990;2:48–50.
44.
Gern L, Rais O. Efficient transmission of Borrelia burgdorferi between cofeeding Ixodes ricinus ticks (Acari: Ixodidae). J Med Entomol. 1996;33(1):189–192.
45.
Labuda M, Alves MJ, Eleckova E, Kozuch O, Filipe AR. Transmission of tick-borne bunyaviruses by cofeeding ixodid ticks. Acta Virol. 1997;41(6):325–328.
46.
Randolph SE. Transmission of tick-borne pathogens between co-feeding ticks: Milan Labuda’s enduring paradigm. Ticks Tick Borne Dis. 2011;2:179–182.
47.
Voordouw MJ. Co-feeding transmission in Lyme disease pathogens. Parasitology. 2015;142(2):290–302. doi:10.1017/S0031182014001486.
48.
Diehl PA, Aeschlimann A, Obenchain FD. Tick reproduction: oogenesis and oviposition. In: Obenchain FD, Galun R, editors. The physiology of ticks. Oxford: Pergamon Press; 1982. p. 277–350.
49.
Gillott C. Male accessory gland secretions: modulators of female reproductive physiology and behavior. Ann Rev Entomol. 2003;48:163–184.
50.
Donohue KV, Khalil SMS, Ross E, Mitchell RD, Roe RM, Sonenshine DE. Male engorgement factor: Role in stimulating engorgement to repletion in the ixodid tick, Dermacentor variabilis. J Insect Physiol. 2009;55:909–918.
51.
Graf J-F. Écologie et éthologie d’Ixodes ricinus L. en Suisse (Ixodoidea: Ixodidae). 3e note: Copulation, nutrition et ponte. Acarologia. 1974;16(4):636–641.
52.
Ioffe-Uspensky I, Uspensky I. The influence of preprandial insemination on feeding and oviposition of Ixodes persulcatus females (Acari: Ixodidae) and some thoughts concerning mating strategies in ticks of the genus Ixodes. Ticks Tick Borne Dis. 2017;8(6):866–871. doi:10.1016/j.ttbdis.2017.07.003.
53.
Graf J-F. Copulation, nutrition et ponte chez Ixodes ricinus L. (Ixodoidea: Ixodidae) – 2e partie. Bull Soc Ent Suisse. 1978;51:241–253.
54.
Balashov YS. Bloodsucking ticks (Ixodoidea) – vectors of diseases of man and animals. Misc Publ Entomol Soc Am. 1972;8:161–376.
55.
Gray JS. Mating and behavioral diapause in Ixodes ricinus L. Exp Appl Acarol. 1987;3(1):61–71. doi:10.1007/BF01200414.
56.
Kiszewski AE, Matuschka FR, Spielman A. Mating strategies and spermiogenesis in ixodid ticks. Annu Rev Entomol. 2001;46:167–182.
57.
Nowak-Chmura, M. Fauna kleszczy (Ixodida) Europy Środkowej. Kraków: Wydawnictwo Naukowe Uniwersytetu Pedagogicznego; 2013. pp. 300.
58.
Bartosik K, Buczek A. Determination of the parameters of the parasitic stage in Ixodes ricinus females. Ann Agric Environ Med. 2013;20(3):441–446.
59.
Rijpkema S, Golubić D, Molkenboer M, Verbeek-De Kruif N, Schellekens J. Identification of four genomic groups of Borrelia burgdorferi sensu lato in Ixodes ricinus ticks collected in a Lyme borreliosis endemic region of northern Croatia. Exp Appl Acarol. 1996;20(1):23–30. doi:10.1007/BF00051474.
60.
Massung RF, Slater K, Owens JH, et al. Nested PCR assay for detection of granulocytic ehrlichiae. J Clin Microbiol. 1998;36(4):1090–1095. doi:10.1128/JCM.36.4.1090-1095.1998.
61.
Blaschitz M, Narodoslavsky-Gföller M, Kanzler M, Stanek G, Walochnik J. Babesia species occurring in Austrian Ixodes ricinus ticks. Appl Environ Microbiol. 2008;74(15):4841–484 6. doi:10.1128/AEM.00035-08.
62.
Renesto P, Gouvernet J, Drancourt M, Roux V, Raoult D. Use of rpoB gene analysis for detection and identification of Bartonella species. J Clin Microbiol. 2001;39(2):430–437. doi:10.1128/JCM.39.2.430-437.2001.
63.
Mysterud A, Hatlegjerde IE, Sorensen OJ. Attachment site selection of life stages of Ixodes ricinus ticks on a main large host in Europe, the red deer (Cervus elaphus). Parasit Vectors. 2014;13(7):510. doi: 10.1186/s13071-014-0510-x.
64.
Ciebiera O, Łopińska A, Gabryś G. Ticks on game animals in the fragmented agricultural landscape of western Poland. Parasitol Res. 2021;120(5):1781–1788. doi:10.1007/s00436-021-07132-9.
65.
Mysterud A, Hügli C, Viljugrein H. Tick infestation on medium–large-sized mammalian hosts: are all equally suitable to Ixodes ricinus adults? Parasit Vectors. 2021;14:254. https://doi.org/10.1186/s13071....
66.
Pavlović I, Jovčevski S, Rogožarski D, Csordás F, Motrović N, Mijatović I, Marčić D, Ilić Ž, Ćirković D, Šekler M, Jovčevski S, Ristić M. Biodiversity of ticks and fleas of dogs in the Western Balkans – Preliminary Examinations. Bull UASVM Vet Med. 2016;73(2):1–4. doi:10.15835/buasvmcn-vm:11344.
67.
Cull B, Pietzsch ME, Hansford KM, Gillingham EL, Medlock JM. Surveillance of British ticks: An overview of species records, host associations, and new records of Ixodes ricinus distribution. Ticks Tick Borne Dis. 2018;9(3):605–614. doi:10.1016/j.ttbdis.2018.01.011.
68.
Zemek,R, Bouman E, Socha R, et al. The effect of feeding status on sexual attractiveness of Ixodes ricinus (Acari: Ixodidae) females. Exp Appl Acarol. 2002;27:137–149. https://doi.org/10.1023/A:1021....
69.
Feldman-Muhsam B, Borut S. Copulation in Ixodid Ticks. J Parasitol. 1971;57(3):630–634.
70.
Feldman-Muhsam B. Observations on the mating behaviour of ticks. In: Sauer JR, Hair JA, editors. Morphology, physiology, and behavioral biology of ticks. Chichester, UK: Ellis Horwood; 1986. p. 217–232.
71.
Pet’ko B. Lyme borreliosis in carpathian region of central Europe – ecological aspect of diagnostics. In: Buczek A, Błaszak C, editors. Arthropods. Arthropods in medicine. Lublin: Koliber; 2002, p. 93–104.
72.
Asman M, Gąsior T, Pająk C, Cuber P, Szilman P, Szilman E, Solarz K. Occupational risk of infections with Borrelia burgdorferi sensu lato, B. burgdorferi sensu stricto, B. garinii and B. afzelii in agricultural workers on the territory of Beskid Zywiecki. In: Buczek A, Błaszak C, editors. Arthropods. The medical and economic importance. Lublin: Akapit; 2012. p. 163–170.
73.
Asman M, Solarz K, Cuber P, et al. Detection of protozoans Babesia microti and Toxoplasma gondii and their co-existence in ticks (Acari: Ixodida) collected in Tarnogórski district (Upper Silesia, Poland). Ann Agric Environ Med. 2015;22(1):80–83.
74.
Asman M, Solarz K, Szilman E, Szilman P, Sikora B, Jakubas-Zawalska J. The occurrence of three tick-borne pathogens in Ixodes ricinus ticks collected from the area of the Kraków-Częstochowa Upland (Southern Poland). Acarologia. 2018;58(4):967–975.
75.
Chunikhin SP, Stefuktina LF, Korolev MB, Reshetnikov IA, Khozinskaia GA. Sexual transmission of the tick-borne encephalitis virus in ixodid ticks (Ixodidae). Parazitologiia. 1983;17(3):214–217.
76.
Alekseev AN, Dubinina HV. Exchange of Borrelia burgdorferi between Ixodes persulcatus (Ixodidae: Acarina) sexual partners. J Med Entomol. 1996a;33(3):351–354.
77.
Alekseev AN, Dubinina HV. Venereal and cannibalistic ways of Borrelia burgdorferi sensu lato exchange between males and females of Ixodes persulcatus (Ixodidae, Acarina). Roczniki Akademii Medycznej w Bialymstoku (1995). 1996b;41(1):103–110.
78.
Gaber MS, Khalil GM, Hoogstraal H. Borrelia crocidurae: Venereal transfer in Egyptian Ornithodorus erraticus ticks. Exp Parasitol. 1982;54:182–184.
79.
Gaber MS, Khalil GM, Hoogstraal H, Aboul-Nasr AE. Borrelia crocidurae localization and transmission in Ornithodoros erraticus and O. savignyi. Parasitology. 1984;88:403–413.
80.
Hayes SF, Burgdorfer W, Aeschlimann A. Sexual transmission of spotted fever group Rickettsiae by infected male ticks: detection of Rickettsiae in immature spermatozoa of Ixodes ricinus. Infect Immun. 1980;27:638–642.
81.
De Silva AM, Fikrig E. Growth and migration of Borrelia burgdorferi in Ixodes ticks during blood feeding. Am J Trop Med Hyg. 1995;53(4):397–404. doi:10.4269/ajtmh.1995.53.397.
82.
Hojgaard A, Eisen RJ, Piesman J. Transmission dynamics of Borrelia burgdorferi s.s. during the key third day of feeding by nymphal Ixodes scapularis (Acari: Ixodidae). J Med Entomol. 2008;45(4):732–736. doi: 10.1603/0022-2585(2008)45[732:TDOBBS]2.0.CO;2. PMID: 18714875.
83.
Dunham-Ems SM, Caimano MJ, Pal U, et al. Live imaging reveals a biphasic mode of dissemination of Borrelia burgdorferi within ticks. J Clin Invest. 2009;119(12):3652–3665. doi:10.1172/JCI39401.
84.
Kurokawa C, Lynn GE, Pedra JHF, Pal U, Narasimhan S, Fikrig E. Interactions between Borrelia burgdorferi and ticks. Nat Rev Microbiol. 2020;18:587–600. doi:10.1038/s41579-020-0400-5.
85.
Lejal E, Moutailler S, Šimo L, Vayssier-Taussat M, Pollet T. Tick-borne pathogenn detection in midgut and salivary glands of adult Ixodes ricinus. Parasit Vectors. 2019;12(1):152. doi:10.1186/s3071-019-3418-7.
86.
Piesman J, Schneider BS, Zeidner NS. Use of quantitative PCR to measure density of Borrelia burgdorferi in the midgut and salivary glands of feeding tick vectors. J Clin Microbiol. 2001;39(11):4145–8. doi:10.1128/JCM.39.11.4145-4148.2001.
87.
Ebel G, Kramer L. Short report: Duration of tick attachment required for transmission of Powassan virus by deer ticks. Am J Trop Med Hyg. 2004;71:268–271.
88.
Richards SL, Langley R, Apperson CS, Watson E. Do tick attachment times vary between different tick-pathogen systems? Environments. 2017;4(2):37. https://doi.org/10.3390/enviro....
89.
Eisen L. Pathogen transmission in relation to duration of attachment by Ixodes scapularis ticks. Ticks Tick Borne Dis. 2018;9(3):535–542. doi:10.1016/j.ttbdis.2018.01.002.
90.
Rocha SC, Velásquez CV, Aquib A, Al-Nazal A, Parveen N. Transmission cycle of tick-borne infections and co-Infections, Animal models and diseases. Pathogens. 2022;11(11):1309. https://doi.org/10.3390/pathog....
91.
Alekseev AN. Tick pathogen interactions: behavior of infected and uninfected ticks (Ixodidae). In: Mitchell R, Horn DJ, Needham GR, Welbourn W, editors. Acarology. Columbus: Biological Survey; 1996. p. 113–15.
92.
Alekseev AN, Dubinina HV. Abiotic parameters and diel and seasonal activity of Borrelia-infected and uninfected Ixodes persulcatus (Acarina: Ixodidae). J Med Entomol. 2000;37(1):9–15. doi:10.1603/0022-2585-37.1.9.
93.
Belova OA, Burenkova LA, Karganova GG. Different tick-borne encephalitis virus (TBEV) prevalences in unfed versus partially engorged ixodid ticks--evidence of virus replication and changes in tick behavior. Ticks Tick Borne Dis. 2012;3(4):240–246. doi:10.1016/j.ttbdis.2012.05.005.
94.
Herrmann C, Gern L. Do the level of energy reserves, hydration status and Borrelia infection influence walking by Ixodes ricinus (Acari: Ixodidae) ticks? Parasitology. 2012;139(3):330–337. doi:10.1017/S0031182011002095.
95.
Pipová N, Peňazziová K, Baňas M, Majláth I, Majláthová V. The Behavior of Rickettsia-positive Dermacentor reticulatus ticks under laboratory conditions. Life (Basel). 2023;13(3):612. doi:10.3390/life13030612.
96.
Buczek W, Buczek A, Witecka J, Asman M. Prevalence of pathogens in sympatric Ixodes ricinus and Dermacentor reticulatus ticks in Eastern Poland and their potential impact on oral-anal contacts between ticks. Ann Agric Environ Med. 2023;30(2):259–265. doi:10.26444/aaem/167427.
97.
Kempf F, de Meeus T, Arnathau C, Degeilh B, McCoy KD. Assortative pairing in Ixodes ricinus (Acari: Ixodidae), the European vector of Lyme Borreliosis. J Med Entomol. 2009;46(3):471–474.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.