Online first
Current issue
Archive
Special Issues
About the Journal
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Board
Editorial Office
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Comparison of the effectiveness of various parasitological methods in detecting nematode eggs in different types of soil
1
Department of Parasitology and Invasive Diseases, National Veterinary Research Institute / State Research Institute, Puławy, Poland
2
Department of Health Biohazards and Parasitology, Institute of Rural Health, Lublin, Poland
3
Department of Plant Nutrition and Fertilization, Institute of Soil Science and Plant Cultivation State Research Institute, Puławy, Poland
4
Department of Soil Science Erosion and Land Protection, Institute of Soil Science and Plant Cultivation State Research Institute, Puławy, Poland
Corresponding author
Jolanta Małgorzata Zdybel
Department of Parasitology and Invasive Diseases, National Veterinary Research Institute State Research Institute, Partyzantow Av., 24-100, Pulawy, Poland
Department of Parasitology and Invasive Diseases, National Veterinary Research Institute State Research Institute, Partyzantow Av., 24-100, Pulawy, Poland
Ann Agric Environ Med. 2023;30(3):425-431
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Natural fertilizers, sewage sludge, digestates, as well as organic fertilizers produced on their basis, can become a source of parasitological contamination of cultivated land. High concentration of invasive forms of parasites in the soil may pose a threat to human and animal health. Therefore, it is necessary to control the hygienic condition of fertilizers and fertilized soils with particular emphasis on parasites. The aim of the study was to compare the effectiveness of methods commonly used for parasitological examination of soil with own methods which were used to develop the standards.
Material and methods:
The study was carried out using samples of sandy soil (SS), horticultural mix soil (HS) and peat-based substrate (PS). Each sample was spiked with 100 dyed Ascaris suum eggs and examined with the use of 6 methods: Vasilkova, Dada, Quinn, and 3 methods according to the Polish Standards (PN-19000, PN- 19005, PN-19006). For each variant, 8 repetitions were made.
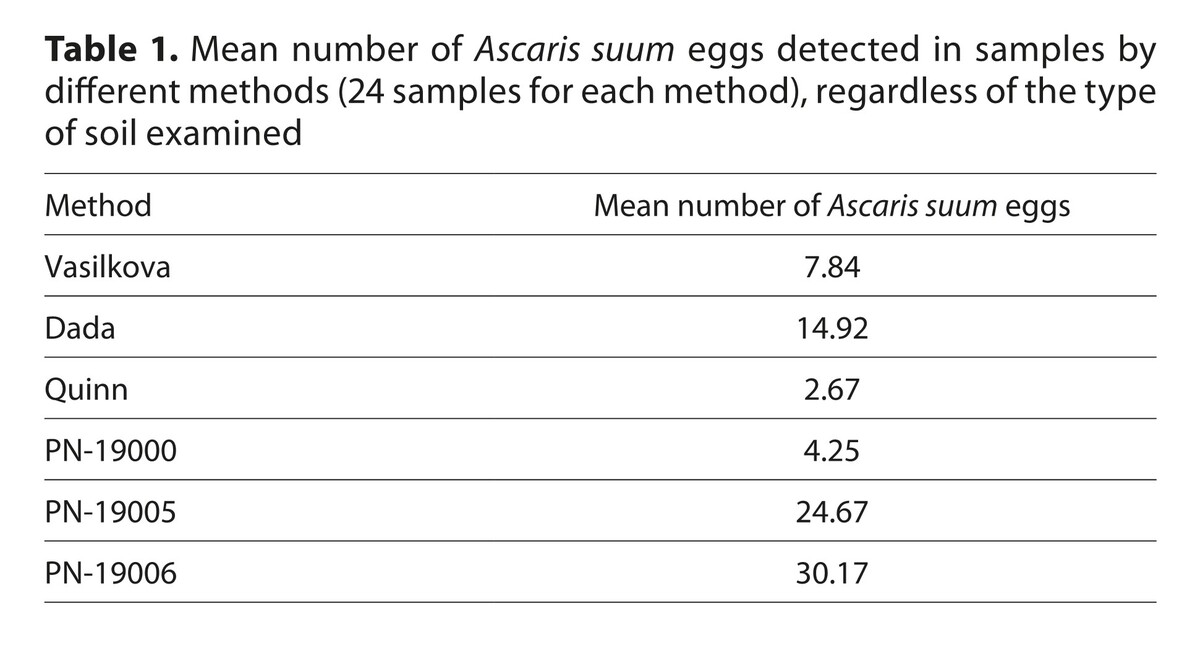
Results:
The largest number of A. suum eggs were found with PN-19006 (mean number of detected eggs was 21.25, 46.50, 23.00 for HS, SS, PS, respectively. Slightly lower results were obtained using PN-19005 – the mean number eggs was 21.25, 36.00, 16.75, respectively. On the other hand, the mean number of A. suum eggs found with the Dada method was about 2–3 times lower than with the PN-19006 – 15.75, 22.50, 6.50 for HS, SS, PS soil, respectively. Other methods were much less effective.
Conclusions:
PN-19006 method turned out to be the most effective in detecting A. suum eggs. This method can be used for parasitological examination of soils and can be the basis for developing a system of methods dedicated to testing different types of soils for the presence of nematode eggs.
Natural fertilizers, sewage sludge, digestates, as well as organic fertilizers produced on their basis, can become a source of parasitological contamination of cultivated land. High concentration of invasive forms of parasites in the soil may pose a threat to human and animal health. Therefore, it is necessary to control the hygienic condition of fertilizers and fertilized soils with particular emphasis on parasites. The aim of the study was to compare the effectiveness of methods commonly used for parasitological examination of soil with own methods which were used to develop the standards.
Material and methods:
The study was carried out using samples of sandy soil (SS), horticultural mix soil (HS) and peat-based substrate (PS). Each sample was spiked with 100 dyed Ascaris suum eggs and examined with the use of 6 methods: Vasilkova, Dada, Quinn, and 3 methods according to the Polish Standards (PN-19000, PN- 19005, PN-19006). For each variant, 8 repetitions were made.
Results:
The largest number of A. suum eggs were found with PN-19006 (mean number of detected eggs was 21.25, 46.50, 23.00 for HS, SS, PS, respectively. Slightly lower results were obtained using PN-19005 – the mean number eggs was 21.25, 36.00, 16.75, respectively. On the other hand, the mean number of A. suum eggs found with the Dada method was about 2–3 times lower than with the PN-19006 – 15.75, 22.50, 6.50 for HS, SS, PS soil, respectively. Other methods were much less effective.
Conclusions:
PN-19006 method turned out to be the most effective in detecting A. suum eggs. This method can be used for parasitological examination of soils and can be the basis for developing a system of methods dedicated to testing different types of soils for the presence of nematode eggs.
ACKNOWLEDGEMENTS
This study was supported by the National Centre for Research and Development (Narodowe Centrum Badań i Rozwój – NCBiR), Grant: GOSPOSTRATEG-III/0061/2020–00 OrgSafety: ‘Introduction of a innovative, cheap and environmentally-friendly method of hygienizing organic waste, enabling its use in fertilization’.
REFERENCES (21)
1.
Rudolfs W, Falk LL, Ragotzkie RA. Contamination of Vegetables Grown in Polluted Soil: III. Field Studies on Ascaris Eggs. Sewage and Industrial Wastes. 1951;23(5):656–60.
2.
Kowalczyk K, Kłapeć T. Contamination of soil with eggs of geohelminths Ascaris spp., Trichuris spp., Toxocara spp. in Poland – potential source of health risk in farmers. Ann Parasitol. 2020;66(4):433–40.
3.
Vasilkova ZG, Gefter VA. Methods for studying soil for helminth eggs. Med Parasitol Parasitic Dis. 1948;(2):139–43.
4.
Dada BJ. A new technique for the recovery of Toxocara eggs from soil. J Helminthol. 1979;53(2):141–4.
5.
PN-Z-19000-4. Soil quality – Assessment of the soil sanitary conditions – detection of eggs of the intestinal parasites Ascaris lumbricoides and Trichuris trichiura. Polish Committee for Standardization; 2001.
6.
Quinn R, Smith HV, Bruce RG, et al. Studies on the incidence of Toxocara and Toxascaris spp. ova in the environment. 1. A comparison of flotation procedures for recovering Toxocara spp. ova from soil. J Hyg (Lond). 1980;84(1):83–9.
7.
Gundłach JL, Sadzikowski AB, Tomczuk K. Contamination by Toxocara spp. eggs of selected urban and rural environments. Med Weter. 1996;52:395–6.
8.
PN-Z-19005:2018-10. Jakość gleby – Ocena stanu sanitarnego materiałów wprowadzanych do gleby – Wykrywanie i oznaczanie ilościowe jaj pasożytów jelitowych z rodzajów Ascaris, Trichuris oraz Toxocara w odwodnionych osadach ściekowych przeznaczonych do wprowadzenia do gleby. Polish Committee for Standardization; 2018.
9.
PN-Z-19006:2023-4. Jakość gleby – Ocena stanu sanitarnego materiałów wprowadzanych do gleby – Wykrywanie jaj pasożytów jelitowych z rodzajów Ascaris, Trichuris oraz Toxocara w nawozach organicznych. Polish Committee for Standardization; 2023.
10.
Zdybel J, Karamon J, Kłapeć T, et al. Negative effect of flocculant (cationic acrylamide) on detectability of the nematode eggs in sewage sludge. J Environ Manage. 2019;231:905–8.
11.
Zdybel J, Karamon J, Różycki M, et al. Characterisation of a new, highly effective method for detecting nematode eggs (Ascaris spp., Toxocara spp., Trichuris spp.) in sewage sludge containing flocculants. Exp Parasitol. 2016;170:198–206.
12.
Cranston I, Teoh PJ, Baker SM, et al. Evaluating the efficacy of a centrifugation-flotation method for extracting Ascaris ova from soil. Trans Roy Soc Trop Med Hyg. 2016;110(7):400–7.
13.
Blaszkowska J, Kurnatowski P, Damiecka P. Contamination of the soil by eggs of geohelminths in rural areas of Lodz district (Poland). Helminthologia. 2011;48(2):67–76.
14.
Oge H, Oge S. Quantitative comparison of various methods for detecting eggs of Toxocara canis in samples of sand. Vet Parasitol. 2000;92(1):75–9.
15.
Willis HH. A simple levitation method for the detection of hookworm ova. Med J Aust. 1921;2.
16.
Ruiz De Ybáñez MR, Garijo M, Goyena M, et al. Improved methods for recovering eggs of Toxocara canis from soil. J Helminthol. 2000;74(4):349–53.
17.
Santarém VA, Magoti LP, Sichieri TD. Influence of variables on centrifuge-flotation technique for recovery of Toxocara canis eggs from soil. Rev Inst Med Trop Sao Paulo. 2009;51(3):163–7.
18.
Kleine A, Janecek E, Waindok P, et al. Flotation and adherence characteristics of Toxocara canis and T. cati and a reliable method for recovering Toxocara eggs from soil. Vet Parasitol. 2016;227:35–41.
19.
Ajala MO, Asaolu SO. Efficiency of the salt flotation technique in the recovery of Ascaris lumbricoides eggs from the soil. J Helminthol. 1995;69(1):1–5.
20.
Loh AG, Israf DA. Tests on the centrifugal flotation technique and its use in estimating the prevalence of Toxocara in soil samples from urban and suburban areas of Malaysia. J Helminthol. 1998;72(1):39–42.
21.
Zdybel J. Assessment of parasitological contamination of municipal sewage sludge in Poland. Doctoral dissertation. National Veterinary Research Institute in Pulawy, Poland, 2016.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.