RESEARCH PAPER
Could the Optiplex Borrelia assay replace the traditional, two-step method of diagnosing Lyme disease?
1
Department of Diagnostic Immunology, Pomeranian Medical University, Szczecin, Poland
2
Immunology Laboratory Independent, Public Clinical Hospital No. 2, Szczecin, Poland
3
Department of Pharmaceutical Microbiology and Microbiological Diagnostic, Medical University, Lodz, Poland
4
Department of Laboratory Medicine, Pomeranian Medical University, Szczecin, Poland
Corresponding author
Iwona Wojciechowska-Koszko
Department of Diagnostic Immunology Pomeranian Medical University in Szczecin, Powstancow Wielkopolskich Av. 72, 70-111, Szczecin, Poland
Department of Diagnostic Immunology Pomeranian Medical University in Szczecin, Powstancow Wielkopolskich Av. 72, 70-111, Szczecin, Poland
Ann Agric Environ Med. 2022;29(1):63-71
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
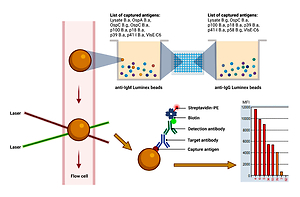
Serological assays for Lyme disease (LD) routinely performed in laboratories often give inconclusive results, thereby making correct diagnosis difficult and delaying treatment. The aim of the study was to assess the usefulness of a commercial Optiplex Borrelia (OB) assay in the serological diagnostics of LD. Based on the results obtained in a previous study on the seroreactivity of the sera of patients with LD to Borrelia spp. antigens using enzyme immunoassays (ELISA) and immunoblotting (IB), the same sera were re-analyzed using the OB assay.
Results:
The assays carried out with the use of OB method showed a statistically significant lower number of positive/borderline results for the presence of IgM antibodies, compared to the ELISA assay. Moreover, statistically lower positive/borderline results were obtained for antibodies in the IgG class with use of the OB method, compared to the IB assay and a two-stage diagnostic protocol (ELISA with IB). The specificity analysis showed that in both the IB and OB assays, anti-OspC IgM and anti-p41 antibodies were detected. Additionally, high positive/borderline values were found in the OB assay for native antigens derived from B. afzelii lysate. The IB assay most frequently detected antibodies against OspC, p39 (BmpA) and VlsE proteins in the IgG class. There were fewer positives/borderlines for anti-p41-I B. afzelii antibodies in the OB assay and a higher number for antigens: VlsE-C6, p18 B. afzelii (DbpA), and p39 B. afzelii (BmpA).
Conclusions:
Answering the question whether the OB assay could replace the traditional, two-step method of LD diagnostics, it can be concluded that it could not. It can be used to diagnose LD only as a complementary assay and not as an optimal and dedicated method of Borrelia spp. infection detection.
Serological assays for Lyme disease (LD) routinely performed in laboratories often give inconclusive results, thereby making correct diagnosis difficult and delaying treatment. The aim of the study was to assess the usefulness of a commercial Optiplex Borrelia (OB) assay in the serological diagnostics of LD. Based on the results obtained in a previous study on the seroreactivity of the sera of patients with LD to Borrelia spp. antigens using enzyme immunoassays (ELISA) and immunoblotting (IB), the same sera were re-analyzed using the OB assay.
Results:
The assays carried out with the use of OB method showed a statistically significant lower number of positive/borderline results for the presence of IgM antibodies, compared to the ELISA assay. Moreover, statistically lower positive/borderline results were obtained for antibodies in the IgG class with use of the OB method, compared to the IB assay and a two-stage diagnostic protocol (ELISA with IB). The specificity analysis showed that in both the IB and OB assays, anti-OspC IgM and anti-p41 antibodies were detected. Additionally, high positive/borderline values were found in the OB assay for native antigens derived from B. afzelii lysate. The IB assay most frequently detected antibodies against OspC, p39 (BmpA) and VlsE proteins in the IgG class. There were fewer positives/borderlines for anti-p41-I B. afzelii antibodies in the OB assay and a higher number for antigens: VlsE-C6, p18 B. afzelii (DbpA), and p39 B. afzelii (BmpA).
Conclusions:
Answering the question whether the OB assay could replace the traditional, two-step method of LD diagnostics, it can be concluded that it could not. It can be used to diagnose LD only as a complementary assay and not as an optimal and dedicated method of Borrelia spp. infection detection.
REFERENCES (35)
1.
Steere AC, Coburn J, Glickstein L. The emergence of Lyme disease. J Clin Invest. 2004; 113(8): 1093–1101. https://doi.org/10.1172/JCI216....
2.
Wojciechowska-Koszko I, Mączyńska I, Szych Z, et al. Serodiagnosis of borreliosis: indirect immunofluorescence assay, enzyme-linked immunosorbent assay and immunoblotting. Arch Immunol Ther Exp (Warsz). 2011; 59(1): 69–77. https://doi.org/10.1007/s00005....
3.
Wojciechowska-Koszko I, Mnichowska-Polanowska M, Kwiatkowski P, et al. Immunoreactivity of polish Lyme disease patient sera to specific Borrelia antigens – Part 1. Diagnostics. 2021; 11(11): 2157. https://doi.org/10.3390/diagno....
4.
Wojciechowska-Koszko I, Kwiatkowski P, Sienkiewicz M, et al. Cross-reactive results in serological tests for borreliosis in patients with active viral infections. Pathogens. 2022; 11(2): 203. https://doi.org/10.3390/pathog....
5.
O Connell S. European Concerted Action on Lyme Borreliosis (EUCALB). Euro Surveill. 1996; 1(3): 23–24.
6.
Robertson J, Guy E, Andrews N, et al. A European multicenter study of immunoblotting in serodiagnosis of Lyme borreliosis. J Clin Microbiol. 2000; 38(6): 2097–2102. https://doi.org/10.1128/JCM.38....
7.
Wilske B. Diagnosis of Lyme borreliosis in Europe. Vector Borne Zoonotic Dis. 2003; 3(4): 215–227. https://doi.org/10.1089/153036....
8.
Aguero-Rosenfeld ME, Wang G, Schwartz I, et al. Diagnosis of Lyme borreliosis. Clin Microbiol Rev. 2005; 18(3): 484–509. https://doi.org/10.1128/CMR.18....
9.
Wilske B, Fingerle V, Schulte-Spechtel U. Microbiological and serological diagnosis of Lyme borreliosis. FEMS Immunol Med Microbiol. 2007; 49(1): 13–21. https://doi.org/10.1111/j.1574....
10.
Hunfeld KP, Kraiczy P. When is the best time to order a Western blot and how should it be interpreted? Curr Probl Dermatol. 2009; 37: 167–177. https://doi.org/10.1159/000213....
11.
Seriburi V, Ndukwe N, Chang Z, et al. High frequency of false positive IgM immunoblots for Borrelia burgdorferi in clinical practice. Clin Microbiol Infect. 2012; 18(12): 1236–1240. https://doi.org/10.1111/j.1469....
12.
Leeflang MMG, Ang CW, Berkhout J, et al. The diagnostic accuracy of serological tests for Lyme borreliosis in Europe: a systematic review and meta-analysis. BMC Infect Dis. 2016; 16: 140. https://doi.org/10.1186/s12879....
13.
Lohr B, Fingerle V, Norris DE, et al. Laboratory diagnosis of Lyme borreliosis: current state of the art and future perspectives. Crit Rev Clin Lab Sci. 2018; 55(4): 219–245. https://doi.org/10.1080/104083....
14.
Theel ES. The past, present, and (possible) future of serologic testing for Lyme disease. J Clin Microbiol. 2016; 54(5): 1191–1196. https://doi.org/10.1128/JCM.03....
17.
Rauer S, Spohn N, Rasiah C, et al. Enzyme-linked immunosorbent assay using recombinant OspC and the internal 14-kDa flagellin fragment for serodiagnosis of early Lyme disease. J Clin Microbiol. 1998; 36(4): 857–861. https://doi.org/10.1128/JCM.36....
18.
Arnaboldi PM, Seedarnee R, Sambir M, et al. Outer surface protein C peptide derived from Borrelia burgdorferi sensu stricto as a target for serodiagnosis of early Lyme disease. Clin Vaccine Immunol. 2013; 20(4): 474–481. https://doi.org/10.1128/CVI.00....
19.
Krupka I, Knauer J, Lorentzen L, et al. Borrelia burgdorferi sensu lato species in Europe induce diverse immune responses against C6 peptides in infected mice. Clin Vaccine Immunol. 2009; 16(11): 1546–1562. https://doi.org/10.1128/CVI.00....
20.
Porwancher RB, Hagerty CG, Fan J, et al. Multiplex immunoassay for Lyme disease using VlsE1-IgG and pepC10-IgM antibodies: improving test performance through bioinformatics. Clin Vaccine Immunol. 2011; 18(5): 851–859. https://doi.org/10.1128/CVI.00....
21.
Probst C, Ott A, Scheper T, et al. N-terminal disulfide-bridging of Borrelia outer surface protein C increases its diagnostic and vaccine potentials. Ticks Tick Borne Dis. 2012; 3(1): 1–7. https://doi.org/10.1016/j.ttbd....
22.
Magnarelli LA, Ijdo JW, Padula SJ, et al. Serologic diagnosis of Lyme borreliosis by using enzyme-linked immunosorbent assays with recombinant antigens. J Clin Microbiol. 2000; 38(5): 1735–1739. https://doi.org/10.1128/JCM.38....
23.
Heikkilä T, Seppälä I, Saxen H, et al. Species-specific serodiagnosis of Lyme arthritis and neuroborreliosis due to Borrelia burgdorferi sensu stricto, B. afzelii, and B. garinii by using decorin binding protein A. J Clin Microbiol. 2002; 40(2): 453–460. https://doi.org/10.1128/JCM.40....
24.
Reed KD. Laboratory testing for Lyme disease: possibilities and practicalities. J Clin Microbiol. 2002; 40(2): 319–324. https://doi.org/10.1128/JCM.40....
25.
Branda JA, Linskey K, Kim YA, et al. Two-tiered antibody testing for Lyme disease with use of 2 enzyme immunoassays, a whole-cell sonicate enzyme immunoassay followed by a VlsE C6 peptide enzyme immunoassay. Clin Infect Dis. 2011; 53(6): 541–547. https://doi.org/10.1093/cid/ci....
26.
Fawcett PT, Rosé CD, Gibney KM, et al. Comparison of immunodot and Western blot assays for diagnosing Lyme borreliosis. Clin Diagn Lab Immunol. 1998; 5(4): 503–506. https://doi.org/10.1128/CDLI.5....
27.
Mogilyansky E, Loa CC, Adelson ME, et al. Comparison of Western immunoblotting and the C6 Lyme antibody test for laboratory detection of Lyme disease. Clin Diagn Lab Immunol. 2004; 11(5): 924–929. https://doi.org/10.1128/CDLI.1....
28.
Ledue TB, Collins MF, Young J, et al. Evaluation of the recombinant VlsE-based liaison chemiluminescence immunoassay for detection of Borrelia burgdorferi and diagnosis of Lyme disease. Clin Diagn Lab Immunol. 2008; 15(12): 1796–1804. https://doi.org/10.1128/CVI.00....
29.
Marangoni A, Sparacino M, Mondardini V, et al. Comparative evaluation of two enzyme linked immunosorbent assay methods and three Western blot methods for the diagnosis of culture-confirmed early Lyme borreliosis in Italy. New Microbiol. 2005; 28(1): 37–43.
30.
Marques AR, Strle F, Wormser GP. Comparison of Lyme disease in the United States and Europe. Emerg Infect Dis. 2021; 27(8): 2017–2024. https://doi.org/10.3201/eid270....
31.
Stanek G, Wormser GP, Gray J, et al. Lyme borreliosis. Lancet. 2012; 379(9814): 461–473. https://doi.org/10.1016/S0140-....
32.
Hauser U, Lehnert G, Wilske B. Validity of interpretation criteria for standardized Western blots (immunoblots) for serodiagnosis of Lyme borreliosis based on sera collected throughout Europe. J Clin Microbiol. 1999; 37(7): 2241–2247. https://doi.org/10.1128/JCM.37....
33.
Mavin S, Evans R, Milner RM, et al. Local Borrelia burgdorferi sensu stricto and Borrelia afzelii strains in a single mixed antigen improves western blot sensitivity. J Clin Pathol. 2009; 62(6): 552–554. https://doi.org/10.1136/jcp.20....
34.
Rauter C, Hartung T. Prevalence of Borrelia burgdorferi sensu lato genospecies in Ixodes ricinus ticks in Europe: a metaanalysis. Appl Environ Microbiol. 2005; 71(11): 7203–7216. https://doi.org/10.1128/AEM.71....
35.
Ivanova L, Christova I, Neves V, et al. Comprehensive seroprofiling of sixteen B. burgdorferi OspC: implications for Lyme disease diagnostics design. Clin Immunol. 2009; 132(3): 393–400. https://doi.org/10.1016/j.clim....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.